Capa Leaves
Blumea balsamifera (Linn.) DC
Asteraceae
DEFINITION
Capa leaves consist of the powder of dried leaves of Blumea balsamifera (Linn.) DC (Asteraceae).
SYNONYM
Baccharis balsamifera Strokes, Baccharis gratissima Blume ex DC., Baccharis salvia Lour., Blumea appendiculata DC., Blumea grandis DC., Blumea zollingeriana (Turcz./ Sch. Bip.) C.B.Clarke, Conyza appendiculata Blume, Conyza balsamifera L., Conyza saxatilisZoll. Ex C.B.Clarke, Pluchea appendiculata (DC.) Zoll. & Mor., Pluchea balsamifera (L.) Less., Blumea balsamifera var. balsamifera, Blumea balsamifera var. microcephala Kitam [ 1 ].
VERNACULAR NAMES
Ngai camphor (English); Sembong, Capa, Telinga Kerbau (Malay); Ai Na Xiang (Chinese) [ 2 ].
CHARACTER
| Colour | Dark yellow-green |
| Odour | Aromatic |
| Taste | Bitter |
IDENTIFICATION
Plant Morphology
B. balsamifera is a perennial herb or subshrub, which rises about 1–3 m in height. Stems are strong, erect and taupe, with longitudinal edges; upper internodes are covered by dense tawny non glandular hair. Leaves are wide, ovoid or oblong-lanceolate at the bottom, 22–25 cm in length, 8–10 cm in width; base is attenuated with petiole, narrow linear appendants of 3–5 pairs on both sides, pubescence above, slight brown or thick yellow-white silky-villous, highlighted below midrib, with lateral veins of 10–15 pairs. Flowers are yellow, with numerous female parts; tubular corolla, receptacle honeycomb, and corolla thin tubulous [ 3 , 4 , 5 ].
Microscopy
The powdered material consists of fragments of wavy epidermal cells, in surface view; group of fibres, isolated; vessels are abundant, consisting of long fragment of scalariform vessel, and fragment of thickened annular vessel; fragments of stomata, mostly isolated; trichomes consist of simple unicellular and multicellular cells.
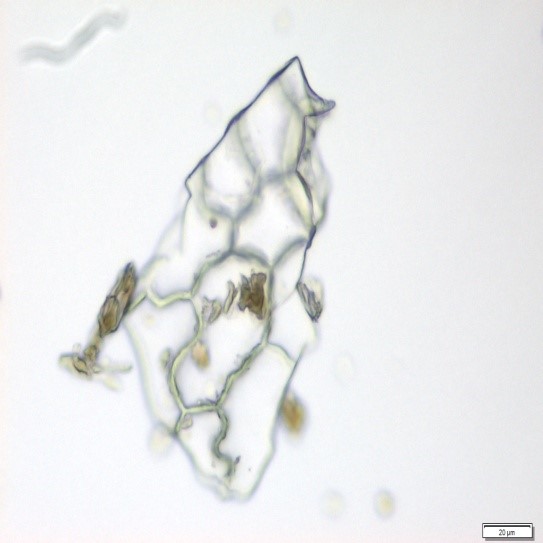

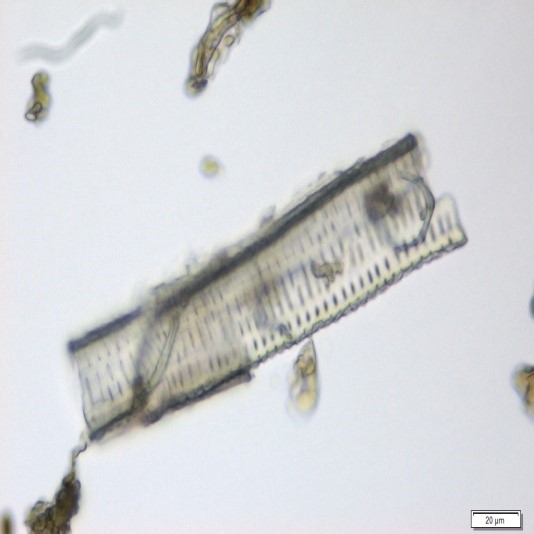
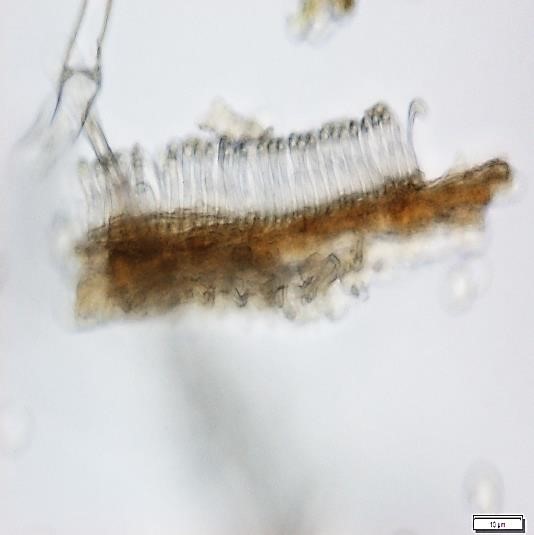
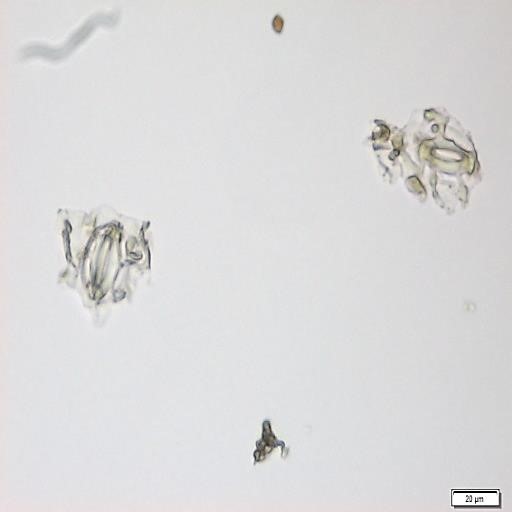
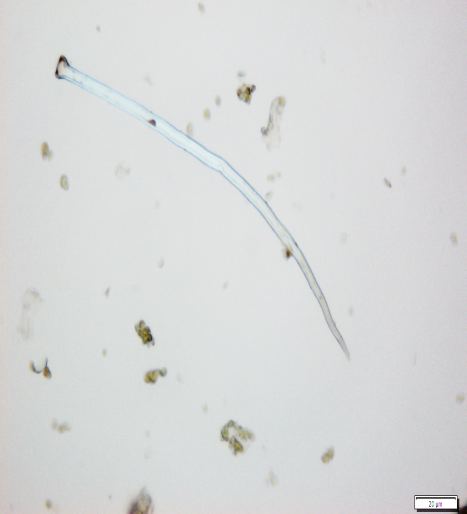
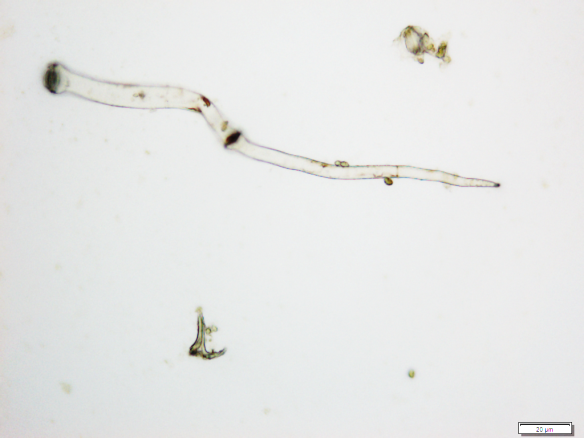
Figure 2 : Microscopic characters of Blumea balsamifera leaves powder of 0.355 mm size. (a) Fragments of epidermal cells (magnification 20x); (b) fragments of fibre (magnification 20x); (c) fragments of scalariform vessel (magnification 20x); (d) fragments of annular vessel (magnification 40x); (e) fragments of stomata (magnification 20x); (f) simple unicellular trichome (magnification 20x); (g) multicellular trichome (magnification 20x). [Scale bars: a, c, e, f, g = 20 µm; b = 50 µm; d = 100 µm]
Colour Tests
Observed colour of solution after treatment with various reagents:
| H2SO4 (conc.) | Green |
| NH4OH (25%) | Green |
Thin Layer Chromatography (TLC)

Figure 3 : TLC chromatogram of eriodictyol (S), ethanol extract of Blumea balsamifera dried leaves powder (L) observed under (a) UV at 254 nm after derivatisation and (b) UV at 366 nm after derivatisation.
| Test Solutions | Weigh about 5.0 g of B. balsamifera dried leaves powder of 0.355 mm in a 100 mL conical flask. Add 25 mL of absolute ethanol and sonicate the mixture for 15 min at room temperature. The mixture is then centrifuged and supernatant is collected. |
| Standard solution | Dissolve eriodictyol standard [CAS no.: 4049-38-1] in ethanol to produce a standard concentration 1.0 mg/mL. |
| Stationary Phase | HPTLC Silica gel 60 F254, 5 x 10cm |
| Mobile phase | Chloroform : methanol ; (8 : 1) (v/v) |
| Application |
|
| Development distance | 8 cm |
| Drying | Air drying |
| Detection |
|
High Performance Liquid Chromatography (HPLC)
| Test solution | Weigh about 2.0 g of B. balsamifera dried leaves powder of 0.355 mm in a 100 mL conical flask and add 25 mL of absolute ethanol. Sonicate the mixture for 15 min at room temperature. The mixture was then centrifuged and supernatant is collected. | ||||||||||||||||||||||||
| Standard solution | Dissolve eriodictyol standard [CAS no: 4049-38-1] in ethanol to produce a standard concentration 0.25 mg/mL. | ||||||||||||||||||||||||
| Chromatographic system |
Detector: 290 nm Column: Column type (150 X 4.6 mm, 3.5 µm) (Zorbax SB-C18 if necessary) Column oven temperature: 25ºC Flow rate: 1.5 mL/min Injection volume: 10 µL |
||||||||||||||||||||||||
| Mobile Phase (gradient mode) |
|
||||||||||||||||||||||||
| System suitability requirement |
Perform at least five replicate injections of the standard solutions (0.25 mg/mL). The requirements of the system suitability parameters are as follow:
|
||||||||||||||||||||||||
| Acceptance criteria |
|
(a)
(b)
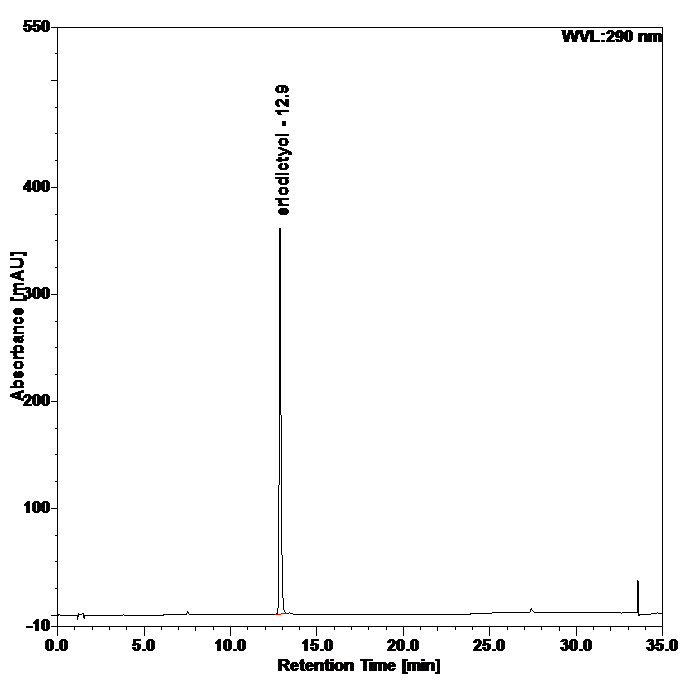
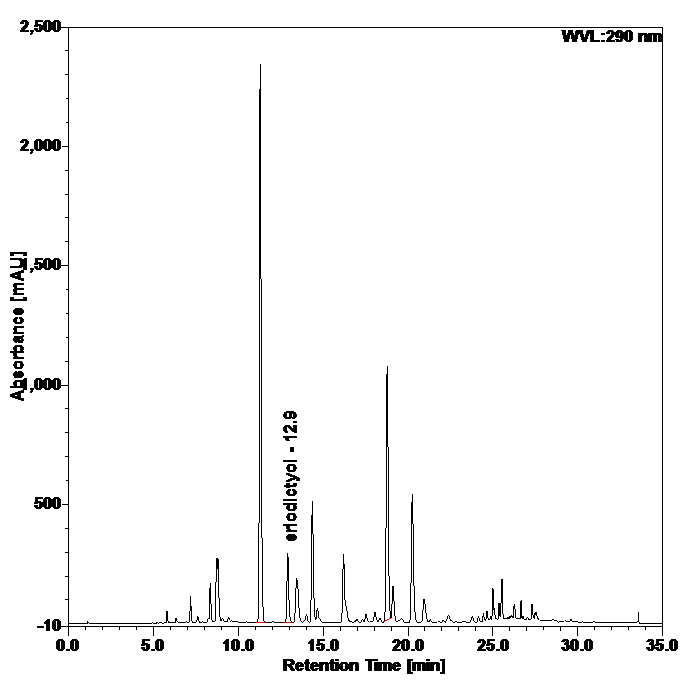
Figure 4 : Whole HPLC chromatogram of (a) eriodictyol standard solution (0.25 mg/mL) at tr = 12.9 min and (b) ethanol extract of Blumea balsamifera dried leaves powder showing peak corresponding to eriodictyol standard solution at tr = 12.9 min.
(a)
(b)
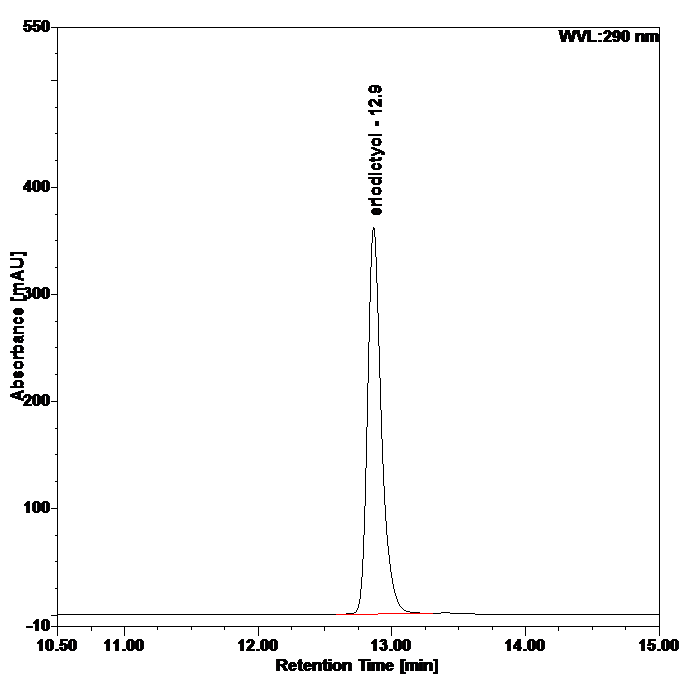
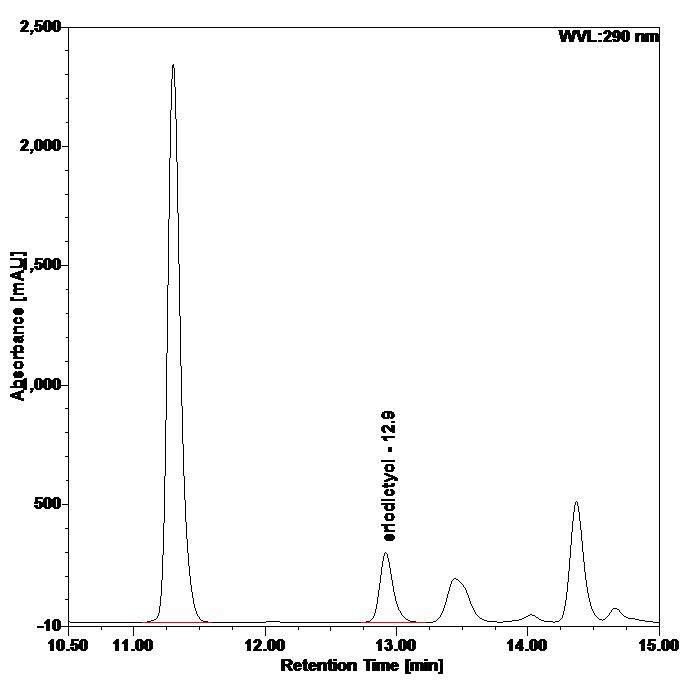
Figure 5 : HPLC chromatogram highlighting the elution region of eriodictyol in (a) eriodictyol standard solution (0.25 mg/mL) and (b) ethanol extract of Blumea balsamifera dried leaves powder showing peak corresponding to eriodictyol standard solution at tr = 12.9 min.
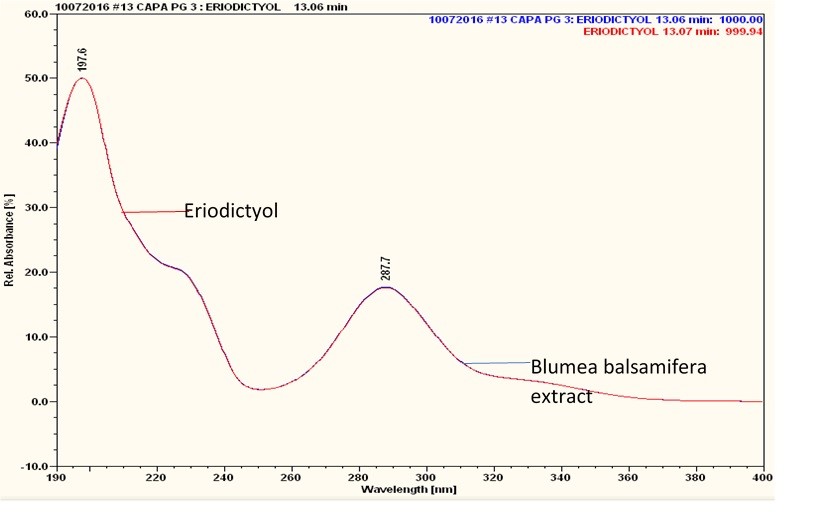
Figure 6 : UV spectrum of eriodictyol standard solution (0.25 mg/mL) and ethanol extract of Blumea balsamifera dried leaves powder.
PURITY TESTS
The purity tests are based on B. balsamifera dried leaves powder of 0.355 mm particle size.
| Foreign Matter |
| Not more than 2% |
| Ash Contents | |
| Total ash | Not more than 13% |
| Acid-insoluble ash | Not more than 1% |
| Loss on Drying |
| Not more than 9% |
| Extractive Values | |
| Water-soluble extracts | |
| Hot method | Not less than 21% |
| Cold method | Not less than 9% |
| Ethanol-soluble extracts | |
| Hot method | Not less than 9% |
| Cold method | Not less than 4% |
SAFETY TESTS
| Heavy Metals | |
| Arsenic | Not more than 5.0 mg/kg |
| Mercury | Not more than 0.5 mg/kg |
| Lead | Not more than 10.0 mg/kg |
| Cadmium | Not more than 0.3 mg/kg |
| Microbial Limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
CHEMICAL CONSTITUENTS
Methanol extract of B. balsamifera dried leaves was found to contain flavonoids (apigenin, dihydroquercetin-7,4’-dimethyl ether, 5,7,3’,5’-tetrahydroxyflavanone, tamarixetin, rhamnetin, luteolin-7-methyl ether, luteolin, quercetin) [ 6 ].
Ethanol extract of B. balsamifera leaves was found to contain flavonoids (eriodictyol), sesquiterpene lactones (blumealactone A, blumealactone B, blumealacone C) [ 7 , 8 ].
Chloroform extract of B. balsamifera leaves was found to contain flavonone (e.g. blumeatin) [ 6 ].
Petroleum ether extract of B. balsamifera dried leaves was found to contain flavonoid (dihydroquercetin-7,4’-dimethylether) [ 6 ].
Essential oil of B. balsamifera dried leaves was found to contain terpenes (borneol, isoborneol, limonene, ocimenecaryophyllene, ledol, phytol, caryophyllene oxide, guaniol), phenol (eugenol, dimethoxydurene) [ 9 ].
MEDICINAL USES
Uses described in folk medicine, not supported by experimental or clinical data
Traditionally the leaves are taken with food as stomachic. The decoction of leaves and roots is used for indigestion, beri-beri and fever. Decoction of the leaves is consumed to treat heavy menstrual bleeding. The boiled leaves are also used to treat lower back pain, joint pain and to bathe women after childbirth [ 10 ].
Biological and pharmacological activities supported by experimental data
Antihyperglycemic activity
Aqueous extract of B. balsamifera leaves inhibited α-glucosidase activity with an 50% inhibition concentration (IC50) value of 0.18 mg/mL compared to positive control, acarbose (IC50 = 0.74 mg/mL) [ 11 ].
Ethyl acetate fraction from ethanol extract of B. balsamifera leaves inhibited α-glucosidase activity with an IC50 value of 0.75 mg/mL compared to acarbose (IC50 = 0.74 mg/mL)
[ 11 ].
Anti-oxidant activity
Methanol extract of B. balsamifera leaves showed significant (p < 0.05) DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity with 50% scavenging concentration (SC50) value of 0.282 ± 0.002 mg/mL compared to α-tocopherol (SC50 = 0.255 ± 0.001 mg/mL) [ 12 ].
Methanol extract of B. balsamifera leaves (0.1 – 1 mg/mL) showed significant (p < 0.05) β-carotene bleaching activity (46.63 – 61.70%) compared to butylated hydroxytoluene, BHT (57.36 – 79.02%) and α-tocopherol (56.25 – 61.67%) using β-carotene-linoleic acid model system [ 13 ].
Chloroform extract of B. balsamifera leaves showed significant (p < 0.05) DPPH radical scavenging activity with SC50 value of 0.294 ± 0.001 mg/mL compared to α-tocopherol with (SC50 = 0.255 ± 0.001 mg/mL) [ 12 ].
Antibacterial activity
Methanol extract of B. balsamifera leaves inhibited the growth of Gram-negative bacteria Escherichia coli, Pseudomonas aeruginosa and Salmonella typhimurium with zone of inhibition ranging from 8 to 9 mm compared to vancomycin (8 – 11 mm), kanamycin (13 – 20 mm), streptomycin (15 – 18 mm) and chloramphenicol (15 – 23 mm) using disc diffusion method [ 14 ].
Methanol extract of B. balsamifera leaves inhibited the growth of Gram-positive acteria Staphylococcus aureus with inhibition zone of 8 mm compared to vancomycin (8 mm), kanamycin (19 mm), streptomycin (18 mm) and chloramphenicol (23 mm) using disc diffusion method [ 14 ].
Dichloromethane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of clinical isolate of Enterobacter cloacae (MSH 003) with inhibition zone of 7 mm compared to gentamicin (7 mm), tetracycline (7 mm), chloramphenicol (11 mm) and penicillin (12 mm) using disc diffusion method [ 15 ].
Dichloromethane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of reference standard strain E. cloacae (DMST 17206) with zone of inhibition of 8 mm compared to gentamicin (10 mm), tetracycline (7 mm), chloramphenicol (7 mm) and penicillin (14 mm) using disc diffusion method [ 15 ].
Dichloromethane extract of B. balsamifera leaves inhibited the growth of reference standard strain E. cloacae (DMST 17206) with minimum inhibitory concentration (MIC) of 9.6 mg/mL using broth macrodilution method [ 15 ].
Hexane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of clinical isolate of S. aureus (MSH 002) with inhibition zone of 6.5 mm compared to gentamicin (21 mm), tetracycline (28 mm), chloramphenicol (22 mm) and penicillin (11 mm) using disc diffusion method [ 15 ].
Hexane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of reference standard strain S. aureus (ATCC 25923) with inhibition zone of 8 mm compared to gentamicin (20 mm), tetracycline (22 mm), chloramphenicol (21 mm) and penicillin (12 mm) using disc diffusion method [ 15 ].
Hexane extract of B. balsamifera leaves inhibited the growth of reference standard strain S. aureus (ATCC 25923) with MIC of 9.6 mg/mL using broth macrodilution method [ 15 ].
Hexane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of clinical isolate of E. cloacae (MSH 003) with inhibition zone of 6.5 mm compared to gentamicin (7 mm), tetracycline (7 mm), chloramphenicol (11 mm) and penicillin (12 mm) using disc diffusion method [ 15 ].
Hexane extract of B. balsamifera leaves (384 µg/disc) inhibited the growth of reference standard strain E. cloacae (DMST 17206) with inhibition zone of 7 mm compared to gentamicin (10 mm), tetracycline (7 mm), chloramphenicol (7 mm) and penicillin (14 mm) using disc diffusion method [ 15 ].
Hexane extract of B. balsamifera leaves inhibited the growth of reference standard strain E. cloacae (DMST 17206) with MIC of 4.8 mg/mL using broth macrodilution method [ 15 ].
Essential oil of B. balsamifera leaves (384 µg/disc) inhibited the growth of Gram-positive, clinical isolate of Bacillus cereus (MSH 001) and S. aureus (MSH 002) with inhibition zone of 12 mm and 19 mm compared to gentamicin (22 and 21 mm), tetracycline (32 and 28 mm), chloramphenicol (24 and 22 mm) and penicillin (12 and 11 mm) using disc diffusion method [ 15 ].
Essential oil of B. balsamifera leaves inhibited the growth of Gram-positive, reference standard strain of B. cereus (ATCC 11778) and S. aureus (ATCC 25923) with MIC of 0.15 and 1.2 mg/mL, respectively using broth macrodilutionmethod [ 15 ].
Antifungal activity
Essential oil of B. balsamifera leaves (384 µg/disc) inhibited the growth of reference standard strain Candida albicans (ATCC 10231) with inhibition zone of 9 mm compared to gentamicin (8 mm), tetracycline (8 mm) and chloramphenicol (9 mm) using disc diffusion method [ 15 ].
Essential oil of B. balsamifera leaves inhibited the growth of reference standard strain Candida albicans (ATCC 10231) with MIC of 1.2 mg/mL using broth macrodilution method
[ 15 ].
Aldolase reductase inhibitory activity
Ethyl acetate fraction of methanol extract of B. balsamifera leaves exhibited aldose reductase inhibitory activities in Sprague Dawley rats (250 – 280 g) lenses with an IC50 value of 0.11 µg/mL compared to 3,3-tetramethyleneglutaric acid (TMG) (IC50 = 0.84 µg/mL) [ 16 ].
Wound healing activity
Essential oil of B. balsamifera leaves diluted in 10% ethanol applied topically once daily to the wounds of adult male and female rats of Sprague Dawley (220 – 250 g) had promoted significant (p < 0.05) burn-wound healing (>90% complete healing) compared to control group (burn ointment) after 21 days of treatment [ 17 ].
Clinical studies
Information and data have not been established.
SAFETY INFORMATION
Preclinical studies (Toxicology studies)
Information and data have not been established.
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Information and data have not been established.
DOSAGE
Information and data have not been established.
STORAGE
Store below 30°C. Protect from light and moisture.
REFERENCES
- The Plant List. [Internet] Blumea balsamifera (L.) DC; 2015 [cited on 7 October 2015]. Available from: http://www.theplantlist.org/tpl1.1/record/gcc-102702.
- Flora of China. [Internet] Blumea balsamifera; [cited 7 October 2015]. Available from: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=200023545.
- Pang Y, Wang D, Fan Z, Chen X, Yu F, Hu X, et al. Blumea balsamifera—A Phytochemical and Pharmacological Review. Molecules. 2014;19(7):9453-77.
- State Administration of Tradisional Chinese Medicine. Chinese materia medica; Scientific and Technical Publishers : Shanghai, China, 1999.
- Chinese Academy of Sciences editorial commision of the flora. Flora Republicae Popularis Sinicae; Sciences Press : Beijing, China, 1979.
- Nessa F, Ismail Z, Mohamed N. Xanthine oxidase inhibitory activities of extracts and flavonoid of the leaves of Blumea balsamifera. Pharmaceutical Biology. 2010; 48(12):1405-1412.
- Huang YL, Zhu TC, Wen YX, Wang HS, Chen YY. Isolation and identification of chemical constituent from Blumea balsamifera. Guihaia. 2010; 30:560-562.
- Fujimoto Y, Soemartono A, Sumatra M. Sesquiterpenelactones from Blumea balsamifera. Phytochemistry. 1988;27(4):1109-11.
- Du P, Zhang XJ, Sun XD. Chemical constituent of volatile oil from Blumea balsamifera (Linn.) DC. InYunan. Chemistry and Industry Forest Product. 2009; 29:76-78.
- Burkill.I.H. A Dictionary of the Economic Product of The Malay Peninsula, Vol.1,1935:334-339.
- Xia Y, Zuo J, Li X, Chen JW. Antihyperglycemic effect of various fractions from Residues of Blumea balsamifera. Chinese Herbal Medicines. 2014;6(2):136-9.
- Nessa F, Ismail Z, Mohamed N, Haris MRHM. Free radical-scavenging activity of organic extracts and of pure flavonoids of Blumea balsamifera DC leaves. Food Chemistry. 2004; 88:243-252.
- Nessa F, Ismail Z, Mohamed N. Antioxidant activity of extracts from the leaves of Blumea balsamifera DC and their major flavooids with the β-carotene–linoleic acid model system. Malaysian Journal of Pharmaceutical Sciences. 2003; 1:51-64.
- Teodora DB, Vilma LV, Melba P, Orlando A, Lianne M, Victoria AL, Jayjay M, Sonny C, Rosemary MG. Phytochemical screening and antibacterial activity of selected medicinal plants of Bayabas, Sablan, Benguet Province, Cordillera A Region, Luzon, Philippines. Indian Journal of Traditional Knowledge. 2012; 11(4): 580-585.
- Sakee U, Maneerat S, Cushnie TPT, De-eknamkul W. Antimicrobial activity of Blumea balsamifera (Lin.) DC. extracts and essential oil. Natural Product Research. 2011; 25(19):1849-56.
- Dong GL, So-Youn M, Changsun C, Eun JC, Hyun YK, Sanghyun L. Analysis of apigenin in Blumea balsamifera Linn DC. and its inhibitory activity against aldolase reductase in rat lens. Journal of Agricultural Chemistry and Environment. 2012; 1:28-33.
- Zuo WF, Yu XP, Kai W, Fu LY, Dan W, Quan Y, Qing SM, Xiao TL, Jin Z, Wen QZ, Li FW. Blumea balsamifera oil for the acceleration of healing of burn injuries. Molecules. 2015; 20:17166-17179.





