Jambu Batu Leaves
Psidium guajava L.
Myrtaceae
Definition
Jambu batu leaves consist of the powder of dried leaves of P. guajava L. (Myrtaceae).
Synonym
Vernacular names
Psidium guajava var. minor Mattos, Psidium igatemyense Barb.Rodr., Myrtus guajava (L.) Kuntze, Psidium prostratum O.Berg, Guajava pumila (Vahl) Kuntze, Psidium pumilum Vahl, Psidium pumilum var. intermedium Blume, Psidium pumilum var. rufescens Blume, Psidium guayava Grisb., Psidium aromaticum Blanco, Psidium fragrans Macfad., Psidium vulgare Rich., Psidium sapidissimum Jacq., Syzygium ellipticum K. Schum. & Lauterb [1, 2].
Character
Identification
Plant Morphology
P. guajava is a shallow-rooted shrub or small tree, up to 10–13 m tall, branching from the base and often producing suckers. Bark is smooth, green to red-brown, peeling off in thin flakes. Young twigs 4–angled and ridged, pubescent. Leaves are simple, opposite, glandular; Leaf blades are elliptical to oblong, 5–15 cm x 3–7 cm, glabrous above, finely pubescent beneath, secondary veins 12–15 on each side of midvein and usually impressed, reticulate veins obvious, base rounded, apex acute to obtuse; Petioles 3–10 mm long. Inflorescences axillary fascicles, each with several staminate flowers and 1 or 2 pistillate flowers. Flowers are solitary or in 2–3-flowered cymes, axillary, approximately 3 cm in diameter; calyx 4–6-lobed, 1–1.5 cm long, irregular, persistent; petals 4–5, white, 1–2 cm long; stamens numerous, 1–2 cm long; ovary 4–5-locular; style 1.5–2 cm long, stigma capitate. Fruits are a berry, globose, ovoid or pyriform, 4–12 cm long, surmounted by the calyx lobes; exocarp green to yellow. Pulp or mesocarp fleshy, white to yellow, with stone cells, sour to sweet, and aromatic. Seeds are usually numerous, embedded in pulp, yellowish, bony, reniform, 3–5 mm long [3, 4].
Microscopy
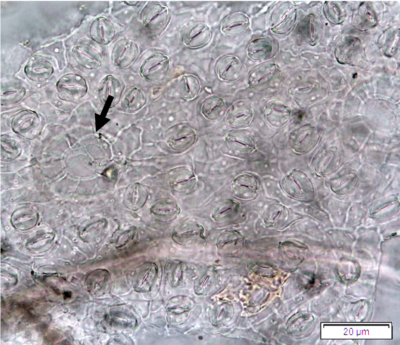
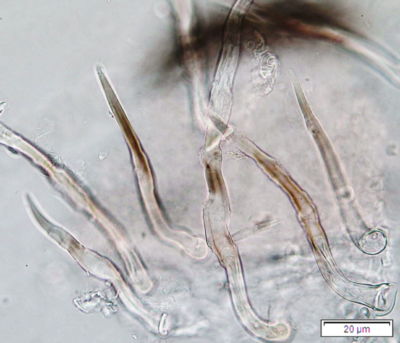
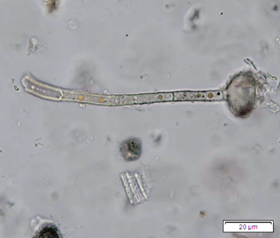
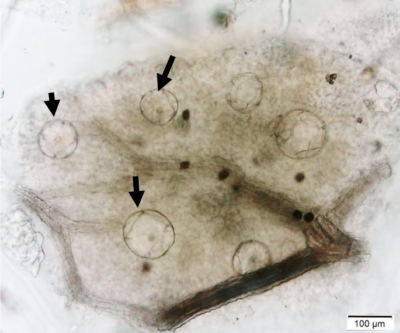
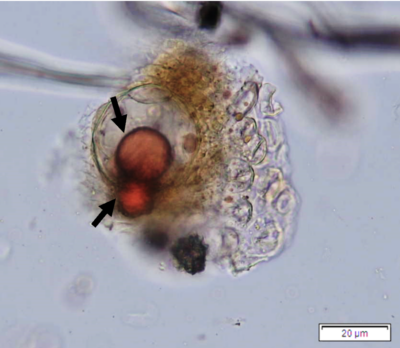
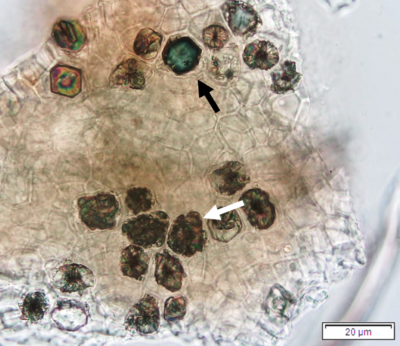
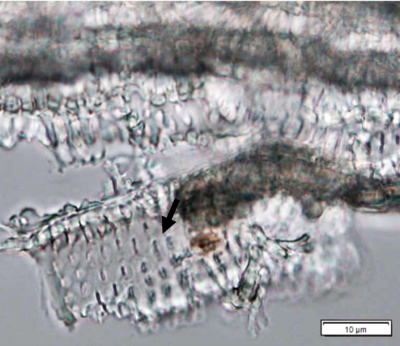
Powdered leaves material consists of adaxial epidermal cells with straight anticlinal walls and abaxial with wavy anticlinal walls, hypostomatic and paracytic stomata; long, simple, unicellular and multicellular with glandular head trichomes and abundant of oil globules in the epidermis cells. Other features observed include the presence of prismatic solitary (rhombus, hexagonal, rectangular, and rod shapes) and druses calcium oxalate crystals; spiral, pitted, and annular vessels; and fibre fragments [7, 8].
Figure 2 : Microscopic characters of P. guajava dried leaves powder of 0.355 mm size. (a) adaxial epidermis cells, straight to curved anticlinal walls (magnification 40x); (b) abaxial epidermis cells, curved to wavy anticlinal walls (magnification 20x); (c) paracytic stomata (arrow) (magnification 40x); (d) long simple, multicellular trichomes (magnification 20x); (e) capitate glandular trichome (magnification 20x); (f) oil globules (arrow) (magnification 4x); (g) oil globules with Sudan red (III) solution (arrow) (magnification 20x); (h) rhombus prism crystals (arrow) (magnification 40x); (i) hexagonal prism crystals (black arrow) and druses crystals (white arrow) (magnification 20x); (j) rectangular (yellow arrow) and rod (black arrow) prism crystals, spiral vessel (red arrow) (magnification 40x); (k) pitted vessel (arrow) (magnification 40x); and (l) annular vessel (magnification 40x). [Scale bars: a, c, e, h, j, k, l = 10 µm; b, d, i, g, j, f = 20 µm]
Chemical Tests
Observation of solution after treatment with various reagents:
| Test for the presence of flavonoids | Colourless |
| Test for the presence of saponin | Stable foams |
| Test for the presence of triterpenoids | Red |
Thin Layer Chromatography (TLC)
| Test Solution | Weigh about 0.5 g of P. guajava dried leaves powder in a 14-mL screw-cap vial. Add 5 mL of methanol and sonicate for 15 min at room temperature. Filter the mixture with a 0.45-µm syringe filter into a HPLC vial. Use the filtrate as a test solution. |
| Standard solution | Dissolved guaijaverin [CAS no.: 22255-13-6] and ellagic acid standards [CAS no.: 476-66-4] in methanol to produce standard concentration of 1 mg/mL solution. |
| Stationary Phase | HPTLC glass silica gel 60 F254, 10 x 10 cm. |
| Mobile phase | Toluene: ethyl acetate: formic acid: water (3 : 3 : 0.8 : 0.2) (v/v/v/v) |
| Application |
|
| Development distance | 8 cm |
| Drying | Oven drying |
| Detection |
|

Figure 3 : TLC chromatogram of guaijaverin (S1), ellagic acid (S2) and methanol extracts of P. guajava dried leaves powder (L) observed under (a) UV at 254 nm before derivatisation with NP-PEG reagent, (b) visible light after derivatisation with NP-PEG reagent, and (c) UV at 366 nm after derivatisation with NP-PEG reagent.
High Performance Liquid Chromatography (HPLC)
| Test Solutions | Weigh about 0.5 g of P. guajava dried leaf powder in a 14-mL screw-cap vial. Add 5 mL of methanol and sonicate for 15 minutes. Filter the mixture with a 0.45-µm syringe filter into a HPLC vial. Use the filtrate as a test solution. | |||||||||||||||||||||
| Standard solution | Dissolved guaijaverin [CAS no.: 22255-13-6] in methanol to produce 1 mg/mL solution. Dilute standard solution in methanol to produce a standard concentration of 0.05 mg/mL solution. | |||||||||||||||||||||
| Chromatographic system | Detector: UV 350 nm Column: C18 column (1.8 μm, 2.1 mm I.D x 100 mm) Column oven temperature: 25°C Flow rate: 0.2 mL/min Injection volume: 3 µL | |||||||||||||||||||||
| Mobile phase(gradient mode) |
| |||||||||||||||||||||
| System suitability requirements | Perform at least five replicate injections of the standard solution (0.05 mg/mL). The requirements of the system suitability parameters are as follow:
| |||||||||||||||||||||
| Acceptance criteria |
|
(b)

(a)

Figure 4 : Whole HPLC chromatogram of (a) guaijaverin standard solution (0.05 mg/mL) at tr = 9.783 min and (b) methanol extract of P. guajava dried leaves powder showing peak corresponding to guaijaverin standard solution at tr = 9.511 min.
(b)

(a)

Figure 5 : HPLC chromatogram highlighting the elution region of guaijaverin of (a) guaijaverin standard solution (0.05 mg/mL) at tr = 9.783 min and (b) methanol extract of P. guajava dried leaves powder showing peak corresponding to guaijaverin standard solution at tr = 9.511 min.

Figure 6 : UV spectrum of guaijaverin standard solution (0.05 mg/mL) and methanol extract of P. guajava dried leaves powder.
Purity Test
The purity tests, except foreign matter test, are based on P. guajava dried leaves powder of 0.355 mm particle size.
| Foreign Matter | |
| Not more than 2 % |
| Ash Contents | |
| Total ash | Not more than 9 % |
| Acid-insoluble ash | Not more than 2 % |
| Loss on Drying | |
| Not more than 11 % |
| Extractive Values | |
| Water-soluble extracts | |
| Hot Method | Not less than 23 % |
| Cold Method | Not less than 15 % |
| Ethanol-soluble extracts | |
| Hot Method | Not less than 25 % |
| Cold Method | Not less than 19 % |
Safety Test
The safety tests are based on P. guajava dried leaves powder of 0.355 mm particle size.
| Heavy Metals | |
| Arsenic | Not More than 5.0mg/kg |
| Mercury | Not More than 0.5 mg/kg |
| Lead | Not More than 10.0 mg/kg |
| Cadmium | Not More than 0.3 mg/kg |
| Microbial Limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
Chemical Constituents
Methanol extract of P. guajava dried leaves was found to contain flavonoids (e.g., rutin, isoquercitrin, quercetin-3-O-β-D-xylopyranoside, quercetin-3-O–α-L-arabinopyranoside, avicularin, quercitrin, quercetin, kaempferol), phenolic (e.g., pyrogallol), sesquiterpenes (e.g., caryophyllene, alloarmadendrene, caryophyllene oxide, epiglobulol, cis-thujopsene), and sesquiterpene hydrocarbons (e.g., α-copaene, β-bisabolene) [9, 10].
Methanol extract of P. guajava dried leaves was found to contain flavonoids (e.g.,rutin, isoquercitrin, quercetin-3-O-β-D-xylopyranoside, quercetin-3-O–α-L-arabinopyranoside, avicularin, quercitrin, quercetin and kaempferol), phenolics (e.g.,pyrogallol), sesquiterpene (e.g., caryophyllene, alloarmadendrene, caryophyllene oxide, epiglobulol, cis-thujopsene), and sesquiterpene hydrocarbon (e.g., α-copaene, β-bisabolene) [11, 12].
Ethanol extract of P. guajava dried leaves was found to contain triterpenoids (e.g., guavanoic acid, guavacoumaric acid), pentacyclic triterpenoids (e.g., 2α-hydroxyursolic acid, asiatic acid), coumaric acids (e.g., jacoumaric acid, isoneriucoumaric acid), and flavonoids (e.g., (+)-psiflavanone A, (-)-psiflavanone A, (+)-psiflavanone B, (-)-psiflavanone B, quercetin, kaempferol, guaijaverin, avicularin, myricetin, hyperin, apigenin) [13, 14, 15].
Essential oil extract of P. guajava dried leaves was found to contain monoterpenes (e.g., limonene, α-pinene, p-cymene, α-thujene, β-myrcene, mentha-2,8 dien-1-ol), terpenes (e.g., β–cis-ocimene, ү-terpinene, β-caryophyllene, α-humulene, β-ocimene, α-terpinene, D-limonene), monocyclic terpene (e.g., α-limonene), terpene alcohols (e.g., linalool, verbenol, terpinen-4-ol, carveol, thujen-3-ol, α-terpineol), terpene ketones (e.g., D-carvone), ester (e.g., butanoic acid-3-hexenyl), cyclic ketone (e.g., verbenone), acetate esters (e.g., carveol acetate, nerol acetate), sesquiterpenes (e.g., caryophyllene, caryophyllene oxide, α-humulene), sesquiterpene hydrocarbons (e.g., α-cubebene, α-murolene, copaene, α-longipinene, β-himachalene, γ-muurolene, α-guaiene, aristolene, cedr-8-ene, δ-cadinene, nerolidol, viridiflorol, γ-cadinene), sesquiterpene alcohols (e.g., ledol, bisabolol), pinene (e.g., 2-norpinene), and essential fatty acids (e.g., linoleic acid, methyl linoleate) [16, 17].
Medicinal Uses
Uses described in folk medicine, not supported by experimental or clinical data
Traditionally, the decoction of the P. guajava leaves is used for diarrhea, stomachache, leucorrhea, swellings, ulcers, gastroenteritis, wounds and also as a vermifuge, vulnerary and a lotion for skin problems [18, 19]. The leaves are chewed as a remedy for toothache; while a decoction of the young leaves and shoots is used to reduce fever and as an antispasmodic bath; and pounded leaves is used for joint pain [19].
Biological and pharmacological activities supported by experimental data
Antioxidant activity
Ethanol extract of P. guajava dried leaves exhibited 2,2-diphenyl-1-picryl hydrazil (DPPH) free radical scavenging activity with 50% inhibition concentration (IC50) value of 89.82 µg/mL compared to ascorbic acid (IC50 = 31.23 µg/mL) and gallic acid (IC50 = 20.08 µg/mL) using DPPH assay [20].
Antidiarrhoeal activity
Ethanol extract of P. guajava dried leaves (750, 500 and 200 mg/kg) was administered orally to Wistar rats (180 g) 6 hours after induction of castor oil to induce diarrhoea. The extract significantly (p < 0.001) decreased the total number of diarrhoeal faeces (7.40 ± 1.14, 11.20 ± 1.48 and 16.60 ± 3.65) compared to loperamide (2.60 ± 0.89) [21].
Antifungal activity
Methanol extract of P. guajava leaves (20 μg/mL) inhibited the growth of Aspergillus niger (inhibition zone = 10 ± 1.0 mm) and Candida albicans (9 ± 1.0 mm) compared to fluconazole (13–14 mm) using paper disc diffusion assay. The minimum inhibitory concentration (MIC) value of the extract against A. nigers and C. albicans is 12.5 mg/mL, respectively [22].
Wound healing activity
Ethanol extract of P. guajava dried leaves (1.0 and 0.5 g/kg body weight) administered orally to alloxan-induced diabetic male Wistar rats (10 g/kg body weight) for a duration of 120 min significantly (p < 0.05) decreased blood glucose level (4.86 ± 1.12 mmol/L and 7.10 ± 0.90 mmol/L, respectively) compared to glibenclamide (2.88 ± 0.34 mmol/L) [21].
Methanol extract of P. guajava dried leaves (1.25%, 2.5%, and 5% formulation) was applied topically on the excised wound (wound size: 350 mm2 and 1–2 mm depth) of Wistar rats (180–200 g body weight, 6–8 weeks old) once daily starting from 48 hours until 20 days. The percentage reduction of wound area on day 20 was 93.8% (1.25% formulation), 100% (2.5% formulation) and 100% (5% formulation) compared to a mixture of bacitracin zinc and neomycin 100% (250 UI/5000 UI) [23].
Anticancer activity
Aqueous and ethanol extracts of P. guajava leaves (150 µg/mL) showed inhibitory activity against the human colon carcinoma cell (HCT116) with inhibition of colony formation of 20.5% and 16.6% compared to suramin (10 mg/mL; 50.72%) using colony formation assay [24].
Betulinic acid isolated from P. guajava leaves (50–800 µg/mL) significantly reduced the viability of HuCCA cells with IC50 of 92.45 µg/mL compared to 5-fluorouracil using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay [25].
Clinical studies
Information and data have not been established.
Safety Information
Preclinical studies (Toxicology study)
Acute toxicity
A dose of 500 mg/kg BW of the methanol extract of P. guajava leaves was applied uniformly to a shaved area of skin Wistar rats were observed for signs of toxicity for 72 hours. Topical application of extract revealed no irritation (no oedema, erythema, and eschar) on healthy skin after 72 hours post-application. The results also showed no ocular irritation effect on the eyes after 48 hours post-application [23].
Oral single dose acute toxicity on adult albino rats (males and females; 100 ± 20 g body weight) using aqueous extract of P. guajava leaves showed no mortality and therefore half lethal dose (LD50) greater than 5,000 mg/kg [26].
Sub-acute toxicity
Aqueous extract of P. guajava leaves orally administrated to adult albino rats (males and females; 100 ± 20 g body weight) daily for 14 days at a dose of 500 mg/kg/ body weight showed no mortality and no sign of hepatotoxicity [26].
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Information and data have not been established.
Dosage
Information and data have not been established.
Storage
Store below 30°C. Protect from light and moisture.
Reference
- World Flora Online. [Internet]. Psidium guajava L.; 2024. [cited on 04 September 2025]. Available from: https://wfoplantlist.org/taxon/wfo-0000284421-2024-12?page=1.
- Malaysia Biodiversity Information System (MyBIS) Database. [Internet]. Psidium guajava L.; 2025. [cited on 04 September 2025]. Available from: https://www.mybis.gov.my/sp/15925.
- Soetopo L. Psidium guajava L..In: Verheij EWM, Coronel RE. (Editors): Plant Resources of South-East Asia. No 2: Edible fruits and nuts. PROSEA Foundation, Bogor, Indonesia. Database record: prota4u.org/prosea. 1991.
- Flora of China @ eFloras. [Internet]. Psidium guajava L.; 1753. [cited on 04 September 2025]. Available from: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=200014806.
- Flora of India @ EfloraofIndia. [Internet]. Psidium guajava L.; 2004. [cited on 04 September 2025]. Available from: https://www.flowersofindia.net/catalog/slides/Guava.html.
- Shaheena S, Chintagunta AD, Dirisala VR, Sampath Kumar NS. Extraction of bioactive compounds from Psidium guajava and their application in dentistry. AMB Express. 2019; 9(208): 2–9.
- Khatijah H, Ruzi AR. Anatomical Atlas of Malaysian Medicinal Plants. Universiti Kebangsaan Malaysia, Bangi, Selangor. 2009, Vol.5: 138–141. ISBN:
- Kementerian Kesehatan Republik Indonesia. Farmakope Herbal Indonesia. Kementerian Kesehatan RI, Jakarta, Indoneisia. 2017, Edisi II: 146 – 149. ISBN: 978-602-416-329-7.
- Liu CW, Wang YC, Lu HC Chiang WD. Optimization of ultrasound-assisted extraction conditions for total phenols with anti-hyperglycemic activity from Psidium guajava leaves. Process Biochemistry. 2014; 49(10): 1601–1605.
- Sampath Kumar NS, Sarbon NM, Rana SS, Chintagunta AD, Prathibha S, Ingilala SK, Jeevan Kumar SP, Sai Anvesh B, Dirisala VR. Extraction of bioactive compounds from Psidium guajava leaves and its utilization in preparation of jellies. AMB Express. 2021; 11(36): 1–9.
- Wang L, Wu Y, Bei Q, Shi K, Wu Z. Fingerprint profiles of flavonoid compounds from different Psidium guajava leaves and their antioxidant activities. Journal of Separation Science. 2017; 40(19): 3817–3829.
- Ashraf A, Sarfraz RA, Rashid MA, Mahmood A, Shahid M, Noor N. Chemical composition, antioxidant, antitumor, anticancer and cytotoxic effects of Psidium guajava leaf extracts. Pharmaceutical Biology. 2016; 54(10): 1971–1981.
- Begum S, Hassan SI, Siddiqui BS, Shaheen F, Nabeel Ghayur M, Gilani AH. Triterpenoids from the leaves of Psidium guajava. Phytochemistry. 2002; 61(4): 399–403.
- Huang J, Li C, Ma J, Xu K, Chen X, Jiang J, Zhang D. Chemical constituents of Psidium guajava leaves and their antibacterial activity. Phytochemistry. 2021; 186, 112746.
- Wang H, Du YJ, Song HC. α-Glucosidase and α-amylase inhibitory activities of guava leaves. Food Chemistry. 2010; 123(1): 6–13.
- Soliman FM, Fathy MM, Salama MM, Saber FR. Comparative study of the volatile oil content and antimicrobial activity of Psidium guajava L. and Psidium cattleianum Sabine leaves. Bulletin of Faculty of Pharmacy, Cairo University. 2016; 54(2): 219–225.
- Ayoola DR, Olonisakin A, Oyeneyin OE. Volatile oil constituents, bioactivity and formulations of essential oil from Psidium guajava. Tropical Journal of Natural Product Research. 2023; 7(7); 3565–3572.
- Burkill IH. A Dictionary of the Economic Products of the Malay Peninsular. 1966, Vol. 2. Ministry of Agriculture and Cooperatives, Kuala Lumpur. 1849.
- Eduardo A. Quisumbing. Medicinal Plants of the Philippines. 1978. Katha Publishing Co. Inc. Quezon City, Philippines. 665–666.
- Braga TV, das Dores RG, Ramos CS, Evangelista FC, da Silva Tinoco LM, de Pilla Varotti F, das Graças Carvalho M, de Paula Sabino A. Antioxidant, antibacterial and antitumor activity of ethanolic extract of the Psidium guajava leaves. American Journal of Plant Sciences. 2014; 5(23): 3492–3500.
- Mazumdar S, Akter R, Talukder D. Antidiabetic and antidiarrhoeal effects on ethanolic extract of Psidium guajava (L.) Bat. leaves in Wister rats. Asian Pacific Journal of Tropical Biomedicine. 2015; 5(1): 10–14.
- Dhiman A, Nanda A, Ahmad S, Narasimhan B. In vitro antimicrobial activity of methanolic leaf extract of Psidium guajava L. Journal of Pharmacy and Bioallied Sciences. 2011; 3(2): 226–229.
- Ekom SE, Tamokou JDD. Methanol leaves extract of Psidium guajava Linn. exhibited antibacterial and wound healing activities. International Journal of Current Microbiology and Applied Sciences. 2018; 7(07): 4008–4023.
- Bronwyn L, Doblin S, Hussein MB, Mansoureh N, Muhammad A, Chu Shan T, AMS Abdul Majid. Anticancer effect of Psidium guajava (Guava) leaf extracts against colorectal cancer through inhibition of angiogenesis. Asian Pacific Journal of Tropical Biomedicine. 2020; 10(7): 293–307.
- Phonarknguen R, Nobsathian S, Assawasuparerk K. Effect of betulinic acid extraction from guava (Psidium guajava Linn.) leaves against human cholangiocarcinoma cells. Asian Pacific Journal of Cancer Prevention. 2022; 23(2): 583 – 90. DOI: 10.31557/APJCP.2022.23.2.583
- Yusuf UA, Ocheje JO. Toxicological studies on the aqueous leaf extract of Psidium guajava in albino rats. Salem University Journal of Life Sciences. 2019; 1(1): 70–76.