Jerangau rhizome
Acorus calamus Linn
Acoraceae
Figure 1 : A. calamus. (a) whole plant with adventitious root; (b) whole plant with rhizome; (c) leaves; (d) spike inflorescence; (e) adventitious root; (f) rhizome; (g) shredded rhizome; and (h) whole plant glasshouse establishment (Photos courtesy of MARDI, 2022).
Definition
Jerangau rhizome consists of the powder of dried rhizome of Acorus calamus Linn. (Acoraceae).
Character
| Colour | Light brown |
| Odour | Aromatic |
| Taste | Bitter and spicy |
Identification
Plant Morphology
A perennial herb up to 80 cm tall, with a grass-like rhizome. Pseudostem erect, glabrous, grooved at one side, and ribbed at the opposite. Leaf simple, bright green, linear to sword-shaped, 40–95 cm long and 10–15 mm wide, thickened at the middle, distichous, acute, and with undulate margins. Inflorescence with a peduncle 20–25 cm long with a very narrow, unequal, short-acuminate spathe, and a green, cylindrical, obtuse spadix 5.0–6.5 cm long by 1.0–1.5 cm broad, densely flowered with bisexual flowers possessing a 6-lobed perianth, six stamens opposite the lobes, flattened filaments ca. 2 mm long, orbicular anthers less than 1 mm long, and a superior, conical, 2–3-loculed ovary. Fruits are small and berry-like, containing three or more seeds, indehiscent. Rootstock stout, with long fibrous roots, spreads horizontally or creeping below the surface of the soil. Seeds conical, 2 mm long [2, 3].
Microscopy
Powdered material consists of round-shaped to oval-shaped grain starches; elongated parenchyma; fragment and annular vessel; collenchyma cells; parenchyma cells, oil globules, and styloid crystal.
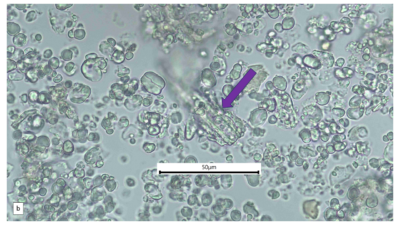
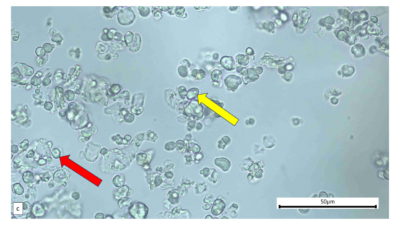
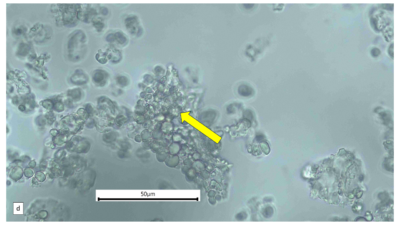
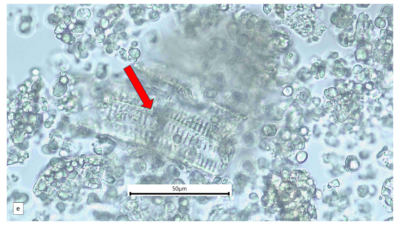
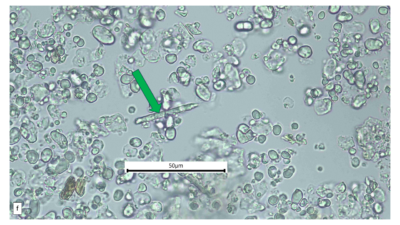
Figure 2 : Microscopic characters of A. calamus rhizome powder of 0.355 mm size. (a) Elongated parenchyma (blue arrow) (magnification 40x); (b) fragment vessel (purple arrow) (magnification 40x); (c) variation of starch grains (oval shape: yellow arrow; round shape: red arrow) (magnification 40x); (d) collenchyma cells (orange arrow) (magnification 40x); (e) annular vessel (pink arrow) (magnification 40x); (f) styloid crystal (green arrow) (magnification 40x); and (g) cluster of oil globules (yellow arrow) (magnification 40x). [Scale bars: a–f = 50 µm; g = 100 µm]
Chemical Tests
Observed colour of solution after treatment for the presence of
| Test for the presence of triterpenes | Bluish green |
| Test for the presence of steroids | White precipitate |
| Test for the presence of saponins | Red |
Thin Layer Chromatography (TLC)
| Test solution | Weigh about 1.0 g of A. calamus dried rhizome powder of 0.355 mm size in a 20-mL glass vial. Add 10 mL of methanol and sonicate the mixture for 30 min. Filter the mixture solution with a 0.22-µm nylon membrane syringe filter into vials. Dilute 10 times by mixing 1 volume of filtrate with 9 volumes of methanol. Use the diluted filtrate as test solution. |
| Standard solution | Dissolve β-asarone [CAS no.: 5273-86-9] in methanol to produce a standard concentration of 1.0 mg/mL. |
| Stationary Phase | HPTLC silica gel plate 60 F254, 10 x 10 cm |
| Mobile phase | Hexane : ethyl acetate; (4 : 1) (v/v) |
| Application | (a) β-asarone solution (S); 3 µL, as a band (b) Methanol extract of A. calamus dried rhizome powder (L); 3 µL, as a band |
| Development distance | 8 cm |
| Drying | Air drying |
| Detection | (a) UV at 254 nm before derivatisation; (b) UV at 366 nm before derivatisation; (c) Visible light after derivatisation with p-anisaldehyde reagent. |

Figure 3 : TLC chromatogram of β-asarone (S) and methanol extract of A. calamus dried rhizome powder (L) observed under (a) UV at 254 nm before derivatisation, (b) UV at 366 nm before derivatisation, and (c) visible light after derivatisation with p-anisaldehyde reagent.
High Performance Liquid Chromatography (HPLC)
| Test solution | Weigh about 1.0 g of A. calamus dried rhizome powder of 0.355 mm size in a 20-mL glass vial. Add 10 mL of methanol and sonicate the mixture for 30 min. Filter the mixture solution with a 0.22-µm nylon membrane syringe filter into vials. Dilute 10 times by mixing 1 volume of the filtrate with 9 volumes of methanol. Use the diluted filtrate as test solution. | |||||||||||||||
| Standard solution | Dissolve β-asarone [CAS no.: 5273-86-9] in methanol to produce a standard concentration of 1.0 mg/mL. | |||||||||||||||
| Chromatographic system | Detector: 280 nm Column: C18 (2.6 µm, 3 mm I.D x 150 mm) Column oven temperature: 30°C Flow rate: 0.6 mL/min | |||||||||||||||
Mobile phase (gradient mode) |
| |||||||||||||||
| Run time | 25 min | |||||||||||||||
| System suitability requirements | Perform at least five replicate injections of the β-asarone (1.0 mg/mL). The requirements of the system suitability parameters are as follow:
| |||||||||||||||
| Acceptance criteria |
|
(b)

(a)

Figure 4 : Whole HPLC chromatogram of (a) β-asarone standard solution (1.0 mg/mL) at tr = 13.032 min and (b) methanol extract of A. calamus dried rhizome powder showing peak corresponding to β-asarone standard at tr = 13.031 min.
(b)

(a)

Figure 5 : HPLC chromatogram highlighting the elution region of (a) β-asarone standard solution (1.0 mg/mL) at tr = 13.032 min and (b) methanol extract of A. calamus dried rhizome powder showing peak corresponding to β-asarone standard at tr = 13.031 min.

Figure 6 : UV spectrum of β-asarone standard solution (1.0 mg/mL) and methanol extract of A. calamus dried rhizome powder.
Purity Test
The purity tests, except foreign matter test, are based on A. calamus dried rhizome powder of 0.355 mm particle size.
| Foreign Matter | |
| Not more than 2% |
| Ash Contents | |
| Total ash | Not more than 5% |
| Acid-insoluble ash | Not more than 1% |
| Water-soluble ash | Not more than 2% |
| Loss on Drying | |
| Not more than 14% |
| Extractive Values | |
| Water-soluble extracts | |
| Hot method | Not less than 13% |
| Cold method | Not less than 7% |
| Ethanol-soluble extracts | |
| Hot method | Not less than 5% |
| Cold method | Not less than 2% |
Safety Test
The safety tests are based on G. species dried plant part powder of 0.355 mm particle size.
| Heavy Metals | |
| Arsenic | Not More than 5.0mg/kg |
| Mercury | Not More than 0.5 mg/kg |
| Lead | Not More than 10.0 mg/kg |
| Cadmium | Not More than 0.3 mg/kg |
| Microbial limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
Chemical Constituents
Medicinal Uses
Uses described in folk medicine, not supported by experimental or clinical data
Biological and pharmacological activities supported by experimental data
Clinical studies
Safety Information
Preclinical studies (Toxicology study)
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Dosage
Information and data have not been established.
Storage
Store below 30°C. Protect from light and moisture.
Reference
- Plants of the World Online. [Internet] Acorus calamus 1753.; [cited on 31 May 2025]. Available from: https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:84009-1#synonyms
- de Padua LS, Bunyaprasphatsara N. and Lemmens RHMJ. Acorus calamus In: de Padua, LS, Bunyapraphatsara N and Lemmens RHMJ. (Edt.). Plant Resources of South-East Asia No. 12: Medicinal and poisonous plants 1. Leiden: Backhuys Publishers 1999; p. 81–85.
- Amit K, Vandana. Medicinal properties of Acorus calamus. Journal of Drug Delivery & 2013;3(3):143–144.
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China. 2015 ed. Vol. 1. Beijing: China Medical Science Press; 2015.
- Matthew, K. M. (1983). The Flora of the Tamilnadu Carnatic. Vol. 3 (2). Diocesan Press, Madras. p. 1683.
- Balakumbahan R, Rajamani K, Kumanan K. Acorus calamus: an overview. Journal of Medicinal Plants Research. 2010;4(25):2740–2745.
- Mc Gaw LJ, Jäger AK, van Staden J. Isolation of β-asarone, an antibacterial and anthelmintic compound, Acorus calamus in South Africa. South African Journal of Botany. 2002;68:31–
- Li J, Zhao J, Wang W, Li L, Zhang L, Zhao XF, Liu QR, Liu F, Yang M, Khan IA, Li SX. New acorane-type sesquiterpene from Acorus calamus Molecules. 2017;22:529.
- Timilsina R, Tandakur P and Pathak I. Biological and chemical studies of essential oil and extracts of rhizome of Acorus calamus Journal of Nepal Chemical Society. 2022;43(1):36–43.
- Satyal P, Paudel P, Poudel A, Dosoky NS, Moriarity DM, Vogler B, Setzer WN. Chemical compositions, phytotoxicity, and biological activities of Acorus calamus essential oils from Nepal. Natural Product Communication. 2013;8(8):1179–1181.
- Liu XC, Zhou LG, Lin ZL and Du SS. Identification of insecticidal constituents of the essential oil of Acorus calamus rhizomes against Liposcelis bostrychophila Molecules. 2013;18:5684–5696.
- Rita WS, Kawuri R, Swantara IMD. The essential oil contents of jeringau (Acorus calamus) rhizomes and their antifungal activity against Candida albicans. Journal of Health Science and Medicine UNUD Journal. 2017;1(1):33–38.
- Srivastava TN, Rajasekharan S, Badola DP, Shah DC: An index of the available medicinal plants usedin Indian system of medicine from Jammu and Kashmirstate. Ancient Science of Life. 1986;6(1):49–63.
- Viswanathan K. Survey on medicinal spices of the Nilgiris. Ancient Science of Life. 1995;14:258–267.
- Jain SP. Tribal remedies from Saranda forest Bihar, India-I. International Journal of Crude Drug Research. 1989;27:29–32.
- Dobriyal RM, Singh GS, Rao KS, Saxena KG. Medicinal plant resources in Chhakinal watershed in the Northwestern Himalaya. Journal of Herbs, Spices & Medicinal Plants. 1997;5(1):15–27.
- Nath P, Yadav AK. Anthelmintic activity of standardized extract from the rhizomes of Acorus calamus (Acoraceae) against experimentally induce cestodiasis in rats. Journal of Intercultural Ethnopharmacology. 2016;5(4):390–395.
- Bari MU, Aslam B, Muhammad Naeem F, Hussain A, Majeed W, Alhindary IA, Abdelrahman SH, Taricone S. The protective effects of Acorus calamus rhizome extracts against rheumatoid arthritis-associated dyslipidemia, oxidative stress, and liver injury in rats. Journal of Applied Animal Research. 2024;52(1).
- Rai AR, Joy T, Poojari M, Pai MM, Massand A, Murlimaju BV. Role of Acorus calamus in preventing depression, anxiety, and oxidative stress in long-term socially isolated rats. Journal of Ayurveda and Integrative Medicine. 2023;14(5):100780.
- Ukkirapandian K, Kayalvizhi E, Udaykumar KP, Kandhi S, Muthulakshmi R. The neuroprotective role of Acorus calamus in developmental and histopathological changes in autism-induced Wistar rats. Cureus. 2022;14(9):e29082.
- Singh BK, Pillai KK, Kohli K, Haque SE. Isoproterenol-induced cardiomyopathy in rats: influence of Acorus calamus A. calamus attenuates cardiomyopathy. Cardiovascular Toxicology. 2011;11(3):263–271.
- Huddar S, Kumar EA. Clinical evaluation of vacha rhizome (Acorus calamus Linn) as Memory Booster in Children. Ayushdhara. 2023;10(5):67–73.
- Shah PD, Ghag M, Deshmukh PB, Kulkarni Y, Joshi SV, Vyas BA, Shah DR. Toxicity study of ethanolic extract of Acorus calamus International Journal of Green Pharmacy. 2012;6(1):29–35.
- European Commission, Scientific Committee on Food. Opinion of the Scientific Committee on Food on β-asarone. Brussels: European Commission; 2002.
- Bhat SD, Ashok BK, Acharya R, Ravishankar B. A comparative acute toxicity evaluation of raw and classically processed rhizomes of vacha (Acorus calamus). Indian Journal of Natural Products and Resources. 2012;3(4):506–511.
- Bai D, Li X, Wang S, Zhang T, Wei Y, Wang Q, Dong W, Song J, Gao P, Li Y, Wang S, Dai L. Advances in extraction methods, chemical constituents, pharmacological activities, molecular targets and toxicology of volatile oil from Acorus calamus angustatus Besser. Frontiers in Pharmacology. 2022;13:1004529.
- S. Food and Drug Administration. Substances Added to Food (formerly EAFUS): Calamus, oil of calamus, extract of calamus [Internet]. Silver Spring (MD): FDA; [cited on 29 December 2025]. Available from: https://hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=FoodSubstances&id=CALAMUSPROHIBITED
- Calamus: Uses, Side Effects, Interactions, Dosage, and Warning [Internet]. [cited on 29 December 2025]. Available from: https://www.webmd.com/vitamins/ai/ingredientmono-778/calamus#precautions