Kari Leaves
Murraya koenigii (L.) Spreng.
Rutaceae
DEFINITION
Kari consists of the dried leaves of Murraya koenigii (L.) Spreng. (Rutaceae).
SYNONYM
Bergera koenigii (L.), Chalcas koenigii (L.) Kurz [ 1 , 2 ].
VERNACULAR NAMES
Curry leaves, curry leaf tree (English); daun kari (Malay); ma jiao ye, ga li cai, ga li ye (Chinese); Kari vempu, Kariya nimpam, Kariveppallai (Tamil) [ 2 ].
CHARACTER
| Colour | Green (powder) |
| Odour | Aromatic |
| Taste | Slightly pungent and bitter [ 3 ] |
IDENTIFICATION
Plant Morphology
M. koenigii is a deciduous shrub or tree up to 4-6 m in height and 15-40 cm in diameter with short trunk, thin smooth grey or brown bark and dense shady crown; bark can be peeled off longitudinally, exposing the white wood underneath. Leaves bipinnately compound, 15-30 cm long, each bearing 11-25 leaflets, alternate on rachis, 2.5-3.5 cm long, ovate lanceolate with an oblique base, margins irregularly creatate, petioles 2-3 mm long. Inflorescence terminal cyme, each bearing 60-90 flowers; calyx 5-lobed, persistent, inferior, green; corolla white, polypetalous, inferior, with 5 petals, lanceolate, length 5 mm; androecium polyandrous, inferior, with 10 stamens, dorsifixed, arranged into circles of five each; stigma bright, sticky; style short; ovary superior. Flowers bisexual, white, funnel-shaped, sweetly scented, stalked, complete, ebracteate, regular, actinomorphic, pentamerous, hypogynous, average diameter of a fully opened flower being 1-1.12 cm. Fruits round to oblong with 1.4 – 1.6 cm long and 1- 1.2 cm in diameter; fully ripe fruits are black with a very shining surface; the number of fruits per cluster varying from 32-80. Seed one in each fruit, 9-11 mm long, 6-8 mm in diameter, colour spinach green [ 3 , 4 , 5 ].
Microscopy
Powdered material consist of simple trichome or broken unicellular covering hairs and multicellular trichome; epidermis adaxial cells with straight to wavy anticlinal wall and attached with stomata; tracheid and solitary calcium oxalate crystal; large pericyclic fibre; cytolith cell; spongy parenchyma and mesophyll; fragment of spiral vessel.





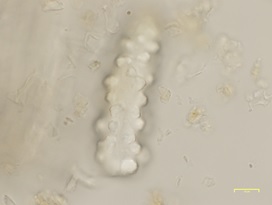


Figure 2 : Microscopic characters of M. koenigii leaves powder; (a) simple trichome or broken unicellular covering hairs (magnification 40x), (b) trichome multicellular (magnification 60x), (c) epidermis adaxial cells with stomata [S] (magnification 20x), (d) calcium oxalate crystal (magnification 60x), (e) fibre (magnification 20x), (f) cystolith cells (magnification 40x), (g) parenchyma cell group (magnification 40x), (h) spiral vessels (magnification 60x). [Scale bars: a-c, e-h = 20 µm; d = 50 µm]
Colour Tests
Observed colour changes on treatment with various reagents:
| H2SO4 (conc.) | Brown |
| HCl (conc.) | Green |
| KOH (5%) | Yellow |
| NH4OH (25%) | Yellow |
Thin Layer Chromatography (TLC)

Figure 3 : TLC profiles of carbazole (S) and extract of M. koenigii leaves powder (L) observed under (a) UV at 254 nm and (b) UV at 366 nm.
| Test Solutions | Weigh about 2 g of M. koenigii dried leaves powder in a screw cap conical flask and add 20 mL of 1% sulphuric in water. Warming the solution on hot plate at 90 °C for 2 minutes with intermittent shaking. Cool the solution at room temperature. Add 5 mL of 2M ammonia to the mixture and shake gently for 3 minutes. Transfer into separating funnel and extract with 10 mL x 3 times of chloroform. Collect the chloroform layer. Add a spatula of anhydrous sodium sulphate to the chloroform mixture and shake. Transfer the chloroform layer into evaporating dish. Evaporate to dryness on water bath. Reconstitute the residue with 2 mL of methanol. Dilute 1 mL in 10 mL of methanol. Filter the solution through a 0.45 µm syringe filter and inject the filtrate into the HPLC column. |
| Standard solution | Dissolve carbazole standard [CAS no.: 86-74-8] in methanol to produce 1000 μg/mL solution (stock). Transfer 1.0 mL of stock solution to make 10 mL with methanol to produce 100 μg/mL |
| Stationary Phase | HPTLC Glass Silica Gel 60 F254, 10 x 10 cm. |
| Mobile phase | Chloroform: methanol: ammonia (95 : 5 : 0.1) (v/v) |
| Application |
|
| Development distance | 8 cm |
| Drying | Dry the plate for 5 minutes with cold air or use the hair dryer. |
| Detection |
|
High Performance Liquid Chromatography (HPLC)
| Test solution | Weigh about 2 g of M. koenigii dried leaves powder in a screw cap conical flask and add 20 mL of 1% sulphuric in water. Warming the solution on hot plate at 90 °C for 2 minutes with intermittent shaking. Cool the solution at room temperature. Add 5 mL of 2M ammonia to the mixture and shake gently for 3 minutes. Transfer into separating funnel and extract with 10 mL x 3 times of chloroform. Collect the chloroform layer. Add a spatula of anhydrous sodium sulphate to the chloroform mixture and shake. Transfer the chloroform layer into evaporating dish. Evaporate to dryness on water bath. Reconstitute the residue with 2 mL of methanol. Dilute 1 mL in 10 mL of methanol. Filter the solution through a 0.45 µm syringe filter and inject the filtrate into the HPLC column. | |||||||||||||||||||||
| Standard solution | Dissolve carbazole standard [CAS no.: 86-74-8] in methanol to produce 1000 μg/mL solution (stock). Transfer 1.0 mL of stock solution to make 10 mL with methanol to produce 100 μg/mL | |||||||||||||||||||||
| Chromatographic system |
Detector: UV 246nm Column: C18 – 250 × 4.6 mm i.d, 5.0 µm particle size. Column oven temperature: 40oC Flow rate: 1.0mL/min. Injection volume: 20µL |
|||||||||||||||||||||
| Mobile Phase (gradient mode) |
|
|||||||||||||||||||||
| System suitability requirement |
Perform at least five replicates injection of the standard solution (1mg/mL). The requirements of the system suitability parameters are as follow:
|
|||||||||||||||||||||
| Acceptance criteria |
|
PURITY TESTS
| Foreign Matter |
| Not more than 2% |
| Ash Contents | |
| Total ash | Not more than 12% |
| Acid-insoluble ash | Not more than 1% |
| Loss on Drying |
| Not more than 10% |
| Extractive Values | |
| Water-soluble extracts | |
| Hot method | Not less than 25% |
| Cold method | Not less than 18% |
| Ethanol-soluble extracts | |
| Hot method | Not less than 15% |
| Cold method | Not less than 9% |
SAFETY TESTS
| Heavy Metals | |
| Arsenic | Not more than 5.0 mg/kg |
| Mercury | Not more than 0.5 mg/kg |
| Lead | Not more than 10.0 mg/kg |
| Cadmium | Not more than 0.3 mg/kg |
| Microbial Limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
CHEMICAL CONSTITUENTS
Ethanol and methanol extract of M. koenigii leaves has been found to contain alkaloids (e.g. carbazole) and flavonoids (e.g. rutin, quercetin, catechin and myricetin) and phenolics (e.g. gallic acid and cinnamic acid) [ 6 , 7 , 8 , 9 ].
Hexane extract of M. koenigii leaves has been found to contain alkaloids (e.g. mahanimbine, isomahanimbine, koenimbidine and murrayacine) [ 10 ].
Chloroform extract of M. koenigii leaves has been found to contain flavonoids (e.g. quercetin) [ 11 ].
MEDICINAL USES
Uses described in folk medicine, not supported by experimental or clinical data
Traditionally used to treat piles, inflammation, itching, fresh cuts, dysentery, vomiting and dropsy [ 12 ]. Different parts of M. koenigii (bark and root) were used for tonic preparation and applied externally to cure eruptions and bites of poisonous animals. Leaves are eaten raw as a cure for dysentery. As decoction with bitters, M. koenigii acts as a reducing fever and is also used in snake bite [ 13 ].
Biological and pharmacological activities supported by experimental data
Antimicrobial activity
Ethanol (90%) extract of M. koenigii leaves (50 mg/mL) inhibited the growth of Staphylococcus aureus with inhibition zones of 16 mm and Escherichia coli (18 mm) compared to vancomycin (16 mm) using disc diffusion assay [ 14 ].
Methanol extract of M. koenigii leaves (50 mg/mL) inhibited the growth of E. coli, Bacillus subtilis and Klebsiella pneumoniae with inhibition zones of 16 mm compared to vancomycin (13 mm) using disc diffusion assay [ 14 ].
Methanol (50%) extract of M. koenigii leaves (5 mg/mL) inhibited the growth of Streptococcus mutans with inhibition zone of 31 mm, Streptococcus sanguinis (27 mm), S. aurues (28 mm), Lactobacillus acidophilus (26 mm) and Lactobacillus casie (28 mm) compared to amoxicillin (12, 14, 12, 13 and 11 mm respectively) using disc diffusion assay [ 15 ].
Antifungal activity
Ethanol (95%) extract of M. koenigii leaves (250 µg/mL) inhibited spore germination of Trichophyton mentagrophytes (50%) and Microsporum gypseum (80%) compare to the control (30% and 40% respectively) [ 16 ].
Anti-inflammatory activity
Methanol (100%) extract of M. koenigii leaves (400 mg/kg) was administered orally to male albino rats (180–200 g) 30 min before induction of paw edema using carrageenan. After four hour, the extracts showed significant (p < 0.05) reduction in paw edema volume (2.65 mL) compared to untreated control group (3.45 mL) [ 17 ].
Methanol extract of M. koenigii leaves (100, 200 and 400 mg/kg/day) was administered orally to male albino rats (25–30 g) 30 min before induction of paw edema using carrageenan. After four hour, the extracts showed significant (p < 0.05) reduction in paw edema volume (0.54-0.33 mL) compared to untreated control group (0.73 mL) [ 18 ].
Anti-oxidant activity
Ethanol:water (1:1) extract of M. koenigii leaves showed anti-oxidant activity with hydroxyl radical scavenging activity inhibition concentration at 50% of growth (IC50) of 10 µg/mL compared to butylated hydroxyanisole (BHA), (IC50 = 40 µg/mL) using deoxyribose assay [ 19 ].
Methanol extract of M. koenigii leaves showed anti-oxidant activity with 1,1-diphenyl-2-picrylhydrazyl (DPPH) scavenging activity with effective concentration at 50% response (EC50) of 6.0 µg/mL compared to trolox (EC50 = 16.2 µg/mL) and butylated hydroxytoluene (BHT) (EC50 = 2.0 µg/mL) using DPPH assay [ 20 ].
Ethanol extract of M. koenigii leaves showed anti-oxidant activity with DPPH scavenging activity with IC50 of 4.10 µg/mL compared to ascorbic acid (IC50 = 2.69 µg/mL) using DPPH assay [ 21 ].
Methanol extract of M. koenigii leaves showed anti-oxidant activity with DPPH scavenging activity with IC50 of 0.028 µg/mL compared to ascorbic acid (IC50 = 0.015 µg/mL) and BHT (IC50 = 0.028 µg/mL) using DPPH assay [ 15 ].
Methanol extract of M. koenigii leaves showed anti-oxidant activity with 2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) radical scavenging activity with IC50 of 0.019 µg/mL compared to BHT (IC50 = 0.013 µg/mL) using ABTS assay [ 15 ].
Methanol extract of M. koenigii leaves (150 µL) showed anti-oxidant activity with Fe2+ reducing ability (3176.98 µmol Fe(II)/g) compared to quercetin (3208.27 µmol Fe(II)/g) using ferric reducing antioxidant potential [ 15 ].
Antiulcer activity
Aqueous extract of M. koenigii leaves (200 and 400 mg/kg) was administered orally as single dose to adult healthy female Wister albino rats (160-200 g/body weight) one hour prior to gastric ulcer induction using ethanol. A significant (p < 0.05) gastro protective activity (58.71%) was observed compared to omeprazole (78.06%). The extract (200 and 400 mg/kg) showed protection index of 47.06% and 58.82% respectively compared to omeprazole 70.59% [ 22 ].
Antitumor activity
Methanol (80%) extract of M. koenigii leaves showed anti-tumour activity against breast cancer cell lines MCF-7 and MDA–MB–231 with IC50 of 37.5 and 15 μg/mL respectively compared to MG-132 (specific inhibitor of the 26S proteasome, carbobenzoxy-Leu-Leu-leucinal) (IC50 = >40 μM and 20 μM respectively) [ 23 ].
Methanol extract of M. koenigii leaves showed anticancer activity against breast cancer cell lines MDA-MB-231 with IC50 of 103 μg/mL compared to tamoxifen (60 μg/mL) [ 9 ].
Methanol (80%) extract of M. koenigii leaves showed anticancer activity against HeLa (breast carcinoma cells), HepG2 (liver carcinoma cells) and LNCaP (prostate adenocarcinoma cells) with IC50 of 4.8, 17.55 and 16.45 µg/mL respectively [ 24 ].
Cytotoxic activity
Ethanol extract of M. koenigii leaves (20 mg/mL) showed 62% cell viability of macrophages RAW264.7 cell line compared to controls (99% cell viability) and dimethyl sulfoxide (125% cell viability) [ 25 ].
Clinical studies
M. koenigii leaves powder supplementation (12 g providing 2.5 g fibre) was carried out for a period of 1 month in 30 non-insulin dependent diabetes mellitus patients (16 males and 14 females) with average age of 59.3 (male) and 57.8 (female). Patients were provided 2 packets of 6 g powder each and consumed 6 g of powder along with lunch and 6 g along with dinner. The 12 g of powder provided 2.4 g protein, 0.4 g fat, 2.5 g crude fibre and 43 calories. M. koenigii leaves supplementation at 12 g per day providing 2.5 g fibre resulted in a significant reduction in fasting blood sugar level (from 148.9 mg/dl to 130.06 mg/dl ) and a non-significant reduction in post-prandial sugar levels (from 206.49 mg/dl to 190.84 mg/dl) at 15 day period [ 26 ].
SAFETY INFORMATION
Preclinical studies (Toxicology studies)
Acute toxicity
Methanol extract of M. koenigii (200-2000 mg/kg body weight) leaves was administered orally as single dose to male Swiss albino rats (145–163 g). After 14 days, moderate toxicity effect was observed (Lethal Dose 50% (LD50) = 316.23 mg/kg body weight) [ 27 ].
Oral single dose acute toxicity study on female Sprague Dawley rats (aged between 8 and 12 weeks old) using aqueous extract of M. koenigii leaves showed no toxic effect on the parameters observed, including behaviours, body weight, food and water intake. All rats were observed for 14 days prior to necropsy. No death was found throughout the study period. Necropsy revealed no significant abnormality. Half lethal dose (LD50) is more than 2,000 mg/kg body weight [ 28 ].
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Information and data have not been established.
DOSAGE
Information and data have not been established.
STORAGE
Store below 30°C. Protect from light and moisture.
REFERENCES
- Darvekar VM, Patil VR, Choudhari AB. Anti-inflammatory activity of Murraya koenigii Spreng on experimental animals. Journal of Natural Product and Plant Resources. 2011;1(1):65-9.
- Plant names. [Internet] Murraya. [cited on 12th Aug 2013]. Available from: http://www.plantnames.unimelb.edu.au/Sorting/Murraya.html.
- Kumar SR, Loveleena D, Godwin S. Medicinal property of Murraya koenigii-A Review. International Research Journal of Biological Sciences 2013;2(9):80-3.
- Handral HK, Pandith A, Shruthi S. A review on Murraya koenigii: multipotential medicinal plant. Asian Journal of Pharmaceutical and Clinical Research. 2012;5(Suppl 4):5-14.
- Parul S, Javed A, Neha B, Honey J, Anuj B. Curry leaves-A Medicinal Herb. Asian Journal of Pharmaceutical and Clinical Research. 2012;2:51-3.
- Mishra J, Yousuf A, Singh RD. Phytochemical investigation and in-vitro antioxidant potential of leaves of Murraya koenigii. International Journal of Integrative Biology. 2009;7(3):171-4.
- Dineshkumar B, Mitra A, Mahadevappa M. Antidiabetic and hypolipidemic effects of mahanimbine (carbazole alkaloid) from Murraya koenigii (rutaceae) leaves. International Journal of Phytomedicine. 2(1).
- Tachibana Y, Kikuzaki H, Lajis NH, Nakatani N. Comparison of antioxidative properties of carbazole alkaloids from Murraya koenigii leaves. Journal of Agricultural and Food Chemistry. 2003;51(22):6461-7.
- Ghasemzadeh A, Jaafar HZ, Rahmat A, Devarajan T. Evaluation of bioactive compounds, pharmaceutical quality, and anticancer activity of curry leaf (Murraya koenigii L.). Evidence-Based Complementary and Alternative Medicine. 2014;2014.10.
- Joshi B, Kamat V, Gawad D. On the structures of girinimbine, mahanimbine, isomahanimbine, koenimbidine and murrayacine. Tetrahedron. 1970;26(5):1475-82.
- Vijayanand S, Wesely E. Phytochemical studies of Melia azadirachta & Murraya koeingi. International Journal of Pharmacy and Pharmaceutical Sciences. 2011;2(5):1298-302.
- Yun-Cheung K, Kam-Hung N, Qian L, Si-Xao Y, Hong-Ta Z, Kin-Fai C, et al. Sources of the anti-implantation alkaloid yuehchukene in the genus Murraya. Journal of Ethnopharmacology. 1986;15(2):195-200.
- Chauhan NS. Medicinal and aromatic plants of Himachal Pradesh: Indus Publishing; 1999.
- Argal M, Kumar S, Choudhary H, Thakkar R, Verma S, Seniya C. The efficacy of Murraya koenigii leaf extract on some bacterial and a fungal strain by disc diffusion method. Journal of Chemical and Pharmaceutical Research. 2011;3:697-704.
- Dwivedi D, Patidar RK, Singh V. Antioxidant and antibacterial potential of Murraya konigii against human cariogenic pathogens. International Journal of Pharmaceutical Sciences and Research. 2012;3:3399-406.
- Jayaprakash A, Ebenezer P. Antifungal activity of curry leaf (Murraya koenigii) extract and an imidazole fungicide on two dermatophyte taxa. 2012.
- Mathur A, Prasad G, Dua V. Anti-inflammatory activity of leaves extracts of Murraya koenigii L. International Journal of Pharma and Bio Sciences. 2011;2(1).
- Gupta S, George M, Singhal M, Sharma GN, Garg V. Leaves extract of Murraya koenigii Linn for anti-inflammatory and analgesic activity in animal models. Journal of Advanced Pharmaceutical Technology and Research. 2011;1(1):68.
- Ningappa MB, Dinesha R, Srinivas L. Antioxidant and free radical scavenging activities of polyphenol-enriched curry leaf (Murraya koenigii L.) extracts. Food chemistry. 2008;106(2):720-8.
- Azlim Almey A, Ahmed Jalal Khan C, Syed Zahir I, Mustapha Suleiman K, Aisyah M, Kamarul Rahim K. Total phenolic content and primary antioxidant activity of methanolic and ethanolic extracts of aromatic plants’ leaves. International Food Research Journal. 2010;17(4).
- Gupta S, Paarakh PM, Gavani U. Antioxidant activity of Murraya Koenigii Linn Leaves. 2009.
- Patidar DK. Anti-ulcer activity of aqueous extract of Murraya koenigii in albino rats. International Journal of Pharma and Bio Sciences. 2011;2(1).
- Noolu B, Ajumeera R, Chauhan A, Nagalla B, Manchala R, Ismail A. Murraya koenigii leaf extract inhibits proteasome activity and induces cell death in breast cancer cells. BMC complementary and alternative medicine. 2013;13(1):7.
- Noolu I, More AI, More XL. Anti-proliferative and proteasome inhibitory activity of Murraya koenigii leaf extract in human cancer cell lines. Discovery Phytomedicine. 2015;2:1-9.
- Naik SK, Mohanty S, Padhi A, Pati R, Sonawane A. Evaluation of antibacterial and cytotoxic activity of Artemisia nilagirica and Murraya koenigii leaf extracts against mycobacteria and macrophages. BMC complementary and alternative medicine. 2014;14(1):87.
- Iyer U, Mani U. Studies on the effect of curry leaves supplementation (Murraya koenigi) on lipid profile, glycated proteins and amino acids in non-insulin-dependent diabetic patients. Plant Foods for Human Nutrition. 1990;40(4):275-82.
- Adebajo A, Ayoola O, Iwalewa E, Akindahunsi A, Omisore N, Adewunmi C, et al. Anti-trichomonal, biochemical and toxicological activities of methanolic extract and some carbazole alkaloids isolated from the leaves of Murraya koenigii growing in Nigeria. Phytomedicine. 2006;13(4):246-54.
- Nor Azlina Z, Emylyn M, Siau TC, Izwah H, Wan Abdul Hakim WL, Wan Mohammad Adham Afiq WZ. Teh BP, Hussin M. Acute oral toxicity study of selected Malaysian medicinal herbs on Sprague Dawley rats. Institute for Medical Research, Ministry of Health; 2015. Report no.: HMRC 11-045/01/MK/L/D.










