Mahkota dewa Leaves
Phaleria macrocarpa (Scheff.) Boerl.
Thymelaeaceae
DEFINITION
Mahkota dewa leaves consist of the powder of dried leaves of Phaleria macrocarpa (Scheff.) Boerl.(Thymelaeaceae).
SYNONYM
Phaleria calantha Gilg, Phaleria papuana Warb. ex. K.Schum & Lauterb., Phaleria papuana var. wichmannii (Valeton)Backer, Phaleria wichmannii Valeton [ 1 ].
VERNACULAR NAMES
God’s crown (English); Mahkota dewa (Malay); Huang kuang (Chinese) [ 2 , 3 ].
CHARACTER
| Colour | Yellowish-green |
| Odour | Odourless |
| Taste | Bitter |
IDENTIFICATION
Plant Morphology
P. macrocarpa is a shrub or an evergreen tree, height ranges from 1–18 m, with 1 m long straight root exuding sap, erect trunk, cylindrical, up to about 15 cm of diameter, smooth or slightly wrinkled brownish bark and dense crown. Leaves simple, oppositely arrangement, elliptic to oblong lanceolate with pointed apex, 7–14 cm long and 3–5 cm broad, coriaceous, dark green in colour; petiole 0.5 cm long. Inflorescence umbel type, peduncle 0.3–2 cm long, borne in the axil of the leaves, carrying 2–5 white sessile flowers. Flowers tubular shaped, 1.5–3.5 cm long, whitish, fragrant, perfect flower with four sepals, four or five petals, 8 stamens and a carpel. Fruits simple, fleshy drupe with one or two seeds, round or elliptical in shape, matured fruit is 3–5 cm in diameter, colour of matured fruit is green and turns to brightly red or maroon when it ripens. Seeds dicotyledonous with exalbuminous seeds type, brown in colour, ovoid, anatropous and exist as 1-2 seeds per fruit [ 4 , 5 ].
Microscopy
The powdered material consists of adaxial epidermal cells with straight to wavy anticlinal wall; fragments of abaxial epidermis with tetracytic stomata; covering trichomes are simple and multicellular with pointed ends; fragments of vascular tissues; druse and solitary crystals are abundant, sometimes found isolated.


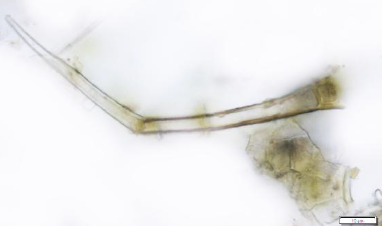
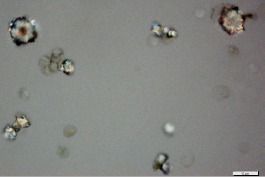

Figure 2 : Microscopic characters of Phaleria macrocarpa leaves powder of 0.355 mm size. (a) Adaxial epidermis cell (magnification 40x); (b) abaxial epidermis cell (magnification 40x); (c) multicellular trichome (magnification 40x); (d) druse calcium oxalate crystals (magnification 40x); (e) solitary prism calcium oxalate crystals (magnification 40x). [Scale bars: a-f = 10 µm]
Colour Tests
Observed colour of solution after treatment with various reagents:
| HCl (conc.) | Green |
| NaOH 5% (w/v) | Green |
Thin Layer Chromatography (TLC)
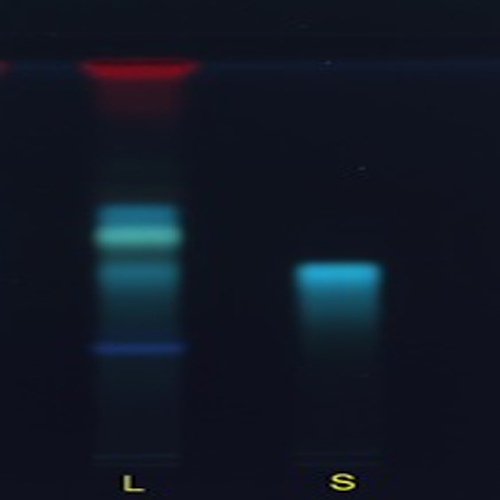
Figure 3 : TLC chromatogram of mangiferin (S), ethanol extract of Phaleria macrocarpa dried leaves powder (L) observed under (a) 366 nm after derivatisation.
| Test Solutions |
Weigh about 2.0 g of P. macrocarpa dried leaves powder of 0.355 mm size in a 100 mL conical flask. Add 50 mL of hexane and shake the mixture for 4 hr at room temperature. Discard the hexane and reconstitute the raw material with 10 mL of ethanol. Sonicate the mixture for 15 min at room temperature. Centrifuge the mixture for 5 min at 13,000 rpm and use the supernatant as test solution. |
| Standard solution |
Dissolve mangiferin standard [CAS no.:4773-96-0] in ethanol to produce a standard concentration 1.0 mg/mL. |
| Stationary Phase | HPTLC Silica gel 60 F254 10×10 cm |
| Mobile phase |
Chloroform : acetone : formic acid : methanol; (35 : 40 : 8 : 15) (v/v/v/v) |
| Application |
|
| Development distance | 8 cm, automatic developing chamber 2 (ADC2) |
| Drying | Air drying |
| Detection |
|
High Performance Liquid Chromatography (HPLC)
| Test solution | Weigh about 1.0 g of P. macrocarpa dried leaves powder of 0.355 mm size in a 50 mL conical flask. Add 5 mL of distilled water and soak the mixture for 30 min at room temperature. Add 20 mL of petroleum ether (b.p 40-60oC) and sonicate the mixture for 15 min at room temperature (cleanup process). Discard the petroleum ether and repeat the cleanup process twice. Allow the raw material to stand for 1 hour at room temperature. Add 10 mL of ethanol and sonicate the mixture for 15 min at room temperature. Centrifuge the mixture and use the supernatant as test solution. | ||||||||||||||||||||||||
| Standard solution | Dissolve mangiferin standard [CAS no: 4773-96-0] in ethanol to produce a standard concentration 0.2 mg/mL. | ||||||||||||||||||||||||
| Chromatographic system |
Detector: 370 nm Column: C18 (150 X 4.6 mm, 3.5 µm) (Zorbax SB-C18 if necessary) Column oven temperature: 25oC Flow rate: 1 mL/min Injection volume: 10 µL |
||||||||||||||||||||||||
| Mobile Phase (gradient mode) |
|
||||||||||||||||||||||||
| System suitability requirement |
Perform at least five replicate injections of the standard solution (0.2 mg/mL). The requirements of the system suitability parameters are as follow:
|
||||||||||||||||||||||||
| Acceptance criteria |
|
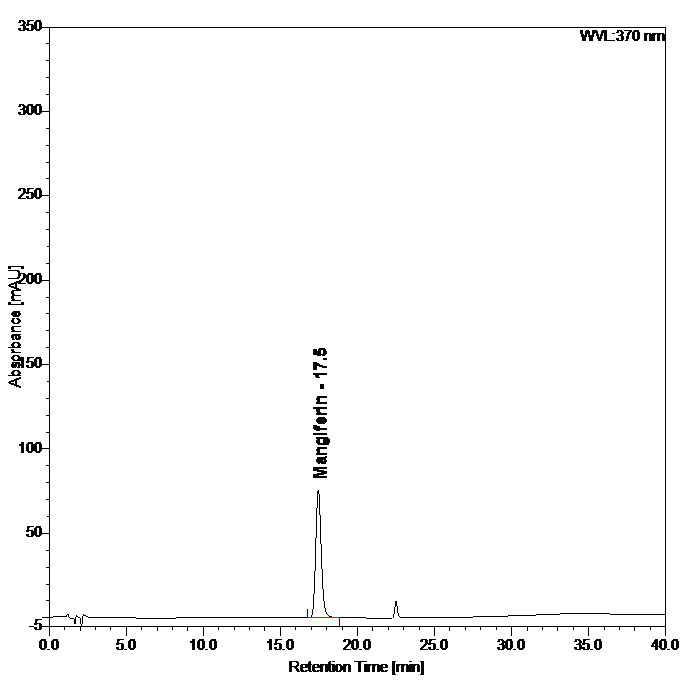
(a)
(b)
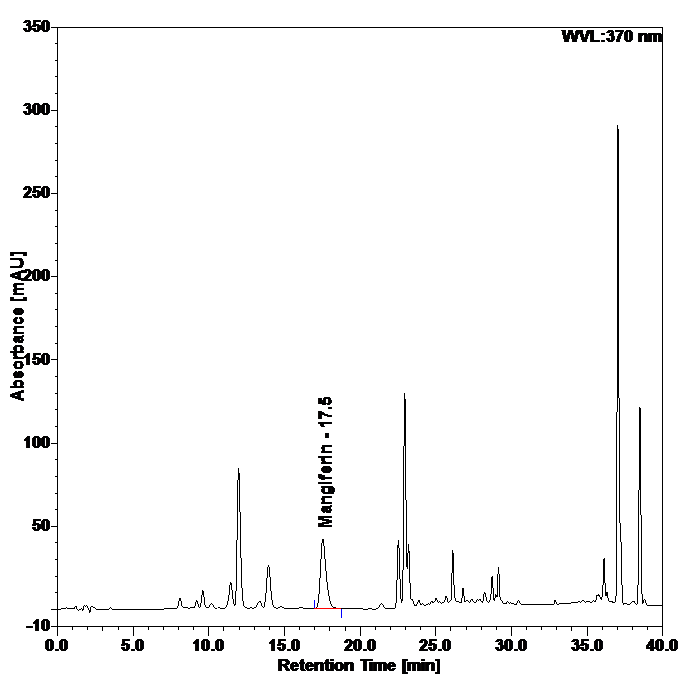
Figure 4 : Whole HPLC chromatogram of (a) mangiferin standard solution (0.2 mg/mL) at tr= 17.5 min and (b) ethanol extract of Phaleria macrocarpa dried leaves powder showing peak corresponding to mangiferin standard solution at tr= 17.5 min.
(a)
(b)
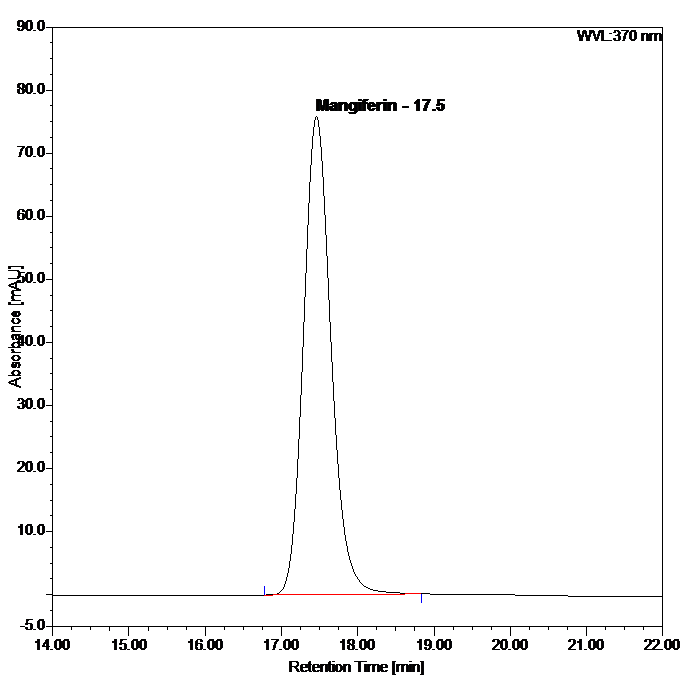
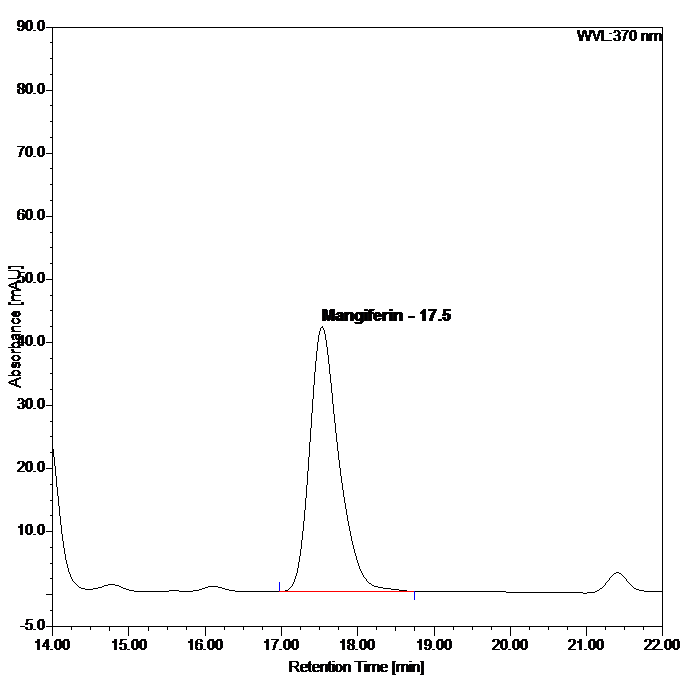
Figure 5 : HPLC chromatogram highlighting the elution region of mangiferin in (a) mangiferin standard solution (0.2 mg/mL) at tr= 17.5 min and (b) ethanol extract of Phaleria macrocarpa dried leaves powder showing peak corresponding to mangiferin standard solution at tr= 17.5 min.
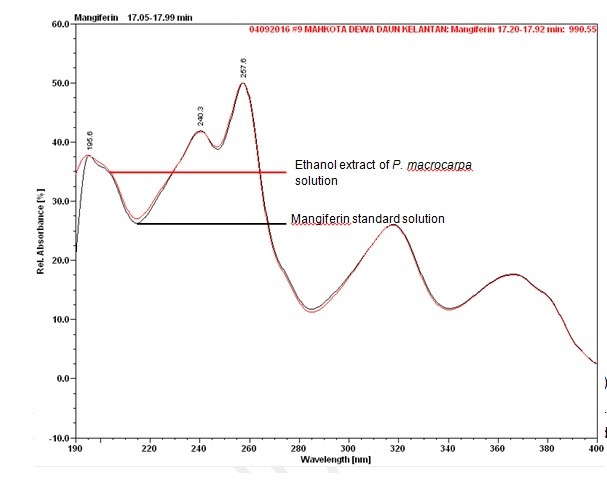
Figure 6 : UV spectrum of mangiferin standard solution (0.2 mg/mL) and ethanol extract of Phaleria macrocarpa dried leaves powder.
PURITY TESTS
The purity tests are based on P. macrocarpa dried leaves powder of 0.355 mm particle size.
| Foreign Matter |
| Not more than 2% |
| Ash Contents | |
| Total ash | Not more than 15% |
| Acid-insoluble ash | Not more than 2% |
| Loss on Drying |
| Not more than 9% |
| Extractive Values | |
| Water-soluble extracts | |
| Hot method | Not less than 27% |
| Cold method | Not less than 22% |
| Ethanol-soluble extracts | |
| Hot method | Not less than 19% |
| Cold method | Not less than 15% |
SAFETY TESTS
The safety tests are based on P. macrocarpa dried leaves powder of 0.355 mm particle size.
| Heavy Metals | |
| Arsenic | Not more than 5.0 mg/kg |
| Mercury | Not more than 0.5 mg/kg |
| Lead | Not more than 10.0 mg/kg |
| Cadmium | Not more than 0.3 mg/kg |
| Microbial Limits | |
| Total bacterial count | Not more than 105 cfu/g |
| Total yeast and mould count | Not more than 104 cfu/g |
| Bile-tolerant gram negative | Not more than 104 cfu/g |
| Specific Pathogens | |
| Salmonella spp. | Absent in 25 g |
| Escherichia coli | Absent in 1 g |
| Staphylococcus aureus | Absent in 1 g |
| Pseudomonas aeruginosa | Absent in 1 g |
CHEMICAL CONSTITUENTS
Methanol extract of P. macrocarpa dried leaves was found to contain benzophenoic glucoside (4,5-dihydroxy, 4’-methoxybenzophenone-3-O-β-D-glucoside; phalerin) [ 6 ].
Ethyl acetate extract of P. macrocarpa dried leaves was found to contain benzophenone aglucon (2,6,4’-trihydroxy-4-methoxybenzophenone) [ 7 ].
n-hexane extract of P. macrocarpa dried leaves was found to contain sterol (β-sitosterol and stigmasterol) [ 8 ].
MEDICINAL USES
Uses described in folk medicine, not supported by experimental or clinical data
Traditionally, the leaves of P. macrocarpa is used for intestinal infection and allergy [ 9 ].
Biological and pharmacological activities supported by experimental data
Antioxidant activity
2,6,4’-trihydroxy-4-methoxybenzophenone isolated from ethyl acetate extract of P. macrocarpa dried leaves (10 – 18 µg/mL) showed DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity with 50% inhibition concentration (IC50) value of 10.57 ± 0.72 µg/mL compared to quercetin (1.2-3.6 µg/mL) with IC50 value of 2.93 ± 2.12 µg/mL) [ 7 ].
Anti-hypercholesterolemic activity
Ethyl acetate extract of P. macrocarpa dried leaves (12.5 µg/mL) increased the hepatic scavenger receptor class B type I (SR-BI) expression to reduce cholesterol levels by 95% compared to positive control rosiglitazone (0.04 µM; 100%) [ 10 ].
Active fraction (CF6) of ethyl acetate extract of P. macrocarpa dried leaves (12.5 µg/mL) increased the SR-BI expression to reduce cholesterol levels by 95% compared to positive control rosiglitazone (0.04 µM; 100%) [ 10 ].
Ethly acetate extract of P. macrocarpa dried powder leaves (0.5 g/kg) was administered orally to female hypercholesterolemia-induced Sprague Dawley rats (7 weeks old; 150 – 180 g) once daily for a duration of 28 days. The extract showed significant (p < 0.05) reduction in the total cholesterol level by 224.9% and increased the HDL cholesterol level by 157% compared to cholesterol treated group [ 10 ].
Clinical studies
Information and data have not been established.
SAFETY INFORMATION
Preclinical studies (Toxicology studies)
Acute Toxicity
Oral single dose acute toxicity study on female Sprague Dawley rats (aged between 8 and 12 weeks old) using aqueous extract of P. macrocarpa leaves showed no toxic effect on the parameters observed, including behaviours, body weight, food and water intake. All rats were observed for 14 days prior to necropsy. No death was found throughout the study period. Necropsy revealed no significant abnormality. Half lethal dose (LD50) is 2,000 mg/kg body weight [ 11 ].
Others (Adverse reaction, contraindication, side effect, warning, precaution)
Information and data have not been established.
DOSAGE
Information and data have not been established.
STORAGE
Store below 30°C. Protect from light and moisture.
REFERENCES
- The Plant List [homepage on the Internet]. Phaleria macrocarpa (Scheff.) Boerl, 2012 [cited on 7 April 2016]. Available from: http://www.theplantlist.org/tpl1.1/record/tro-50315226
- Anggraini T, Lewandowsky P. The exotic plants of Indonesia: Mahkota dewa (Phaleria macrocarpa), sikaduduak (Melastoma malabathricum Linn) and mengkudu (Morinda citrifolia) as potent antioxidant sources. International Journal on Advanced Science Engineering Information Technology. 2015;5(2):115-118.
- Hai Kou Yi Yu Cao Mu Health Food Co. Ltd [Internet] Phaleria macrocarpa; 2015 [cited on 20 December 2016]. Available from: http://www.hkyycm.com/news2.aspx?cid=1&id=21
- Rabia A, Mohammad ZBA, Aidiahmad D, Amirin S, Muhammad IU. Phytochemistry and medicinal properties of Phaleria macrocarpa (Scheff.) Boerl. extracts. Pharmacognosy Review. 2013; 7(13):73-80.
- Hasimah A, Nor Nafizah MN, Norhayati D, Hasnah I. The inflorescence and infructescence morphology of Phaleria macrocarpa (Boerl.) Scheff. Journal of Science and Mathematics. 2013;5:67-72.
- Hartati WMS, Mubarika S, Gandjar I, Hamann MT, Rao KV, Wahyuono S. Phalerin, a new benzophenoic glucoside isolated from the methanolic extract of Mahkota Dewa [Phaleria macrocarpa (Scheff).Boerl.] leaves. Majalah Farmasi Indonesia. 2005;16(1):51-57.
- Susilawati, Matsjeh S, Pranowo HD, Anwar C. Antioxidant activity of 2,6,4’-trihydroxy-4-methoxybenzophenone from ethyl acetate extract of leaves of mahkota dewa (Phaleria macrocarpa (Scheff.) Boerl.). Indonesian Journal of Chemistry. 2011;11(2):180-185.
- Othman SNAM, Sarker SD, Talukdar AD, Ningthoujam SS, Khamis S, Basar N. Chemical constituents and antibacterial activity of Phaleria macrocarpa (Scheff.) Boerl. International Journal of Pharmaceutical Sciences and Research. 2014; 5(8):3157-3162.
- Harmanto N. Conquering disease in unison with Mahkota dewa. North Jakarta: Ir. Harmantop, 2003; p.14.
- Yosie A, Sifzizul TMT, Habsah M, Jasnizat S, Desy FS, Guat SC, Effendy MAW. Phaleria macrocarpa Boerl. (Thymelaeaceae) leaves increase SR-BI xpression and reduce cholesterol levels in rats fed a high cholesterol diet. Molecules. 2015; 20:4410-4429.
- Norzahirah A, Umi Rubiah SZ, Bazilah J, Nurul Syahira S, Dalnitha Lea N, Tavinraj R, Teh BP. Acute oral toxicity study of selected Malaysian medicinal herbs on Sprague Dawley rats. Institute for Medical Research, Ministry of Health ; 2019. Reportno.: NON-GLP/2019/07/01.







